Takotsubo cardiomyopathy (TC), also known as transient left ventricular apical ballooning syndrome, stress-induced cardiomyopathy and broken heart syndrome, is characterized by transient left ventricular dysfunction in the absence of obstructive coronary artery disease.
It was first described in 1990 in Japan, and gained worldwide recognition following the publication of several series of case reports. Its prevalence is estimated to be 1.7–2.2% of suspected acute coronary syndromes. Although TC has been progressively better characterized, certain aspects remain to be clarified, and it is still under study.
In this article, we report a series of ten cases of TC admitted to a cardiology department, and compare the clinical, laboratory, electrocardiographic and imaging characteristics, therapeutic regimens and follow-up of these patients with those described in the latest scientific reviews.
A Miocardiopatia de Takotsubo (MCT), mais recentemente denominada Acinesia/Discinesia Apical Transitória do Ventrículo Esquerdo, Miocardiopatia Induzida por Stress, Síndrome do Coração Roto, caracteriza-se pela alteração reversível da contractilidade segmentar do ventrículo esquerdo (VE) na ausência de lesão coronária significativa.
Foi descrita pela primeira vez em 1990, no Japão, assistindo-se posteriormente a um considerável reconhecimento mundial, através da publicação de várias séries de casos clínicos, calculando-se uma prevalência de 1,7 a 2,2% das situações em que existe suspeição clínica de síndrome coronário aguda (SCA). Apesar da progressiva caracterização, existem pontos a esclarecer, pelo que a MCT é ainda actualmente objecto de estudo.
Neste artigo, descreve-se uma série de dez casos de MCT que ocorreram num Serviço de Cardiologia, com o objectivo de correlacionar as características clínicas, laboratoriais, electrocardiográficas, imagiológicas, complicações, terapêutica instituída e follow-up destes doentes, com as descritas nas revisões científicas actuais.
Takotsubo cardiomyopathy (TC) is characterized by acute onset of akinesia/dyskinesia of the apical and mid segments of the left ventricle (LV), usually triggered by stress. It is included in the wide range of differential diagnoses of acute coronary syndrome (ACS), with reversibility and absence of significant coronary lesions being its main distinguishing features.
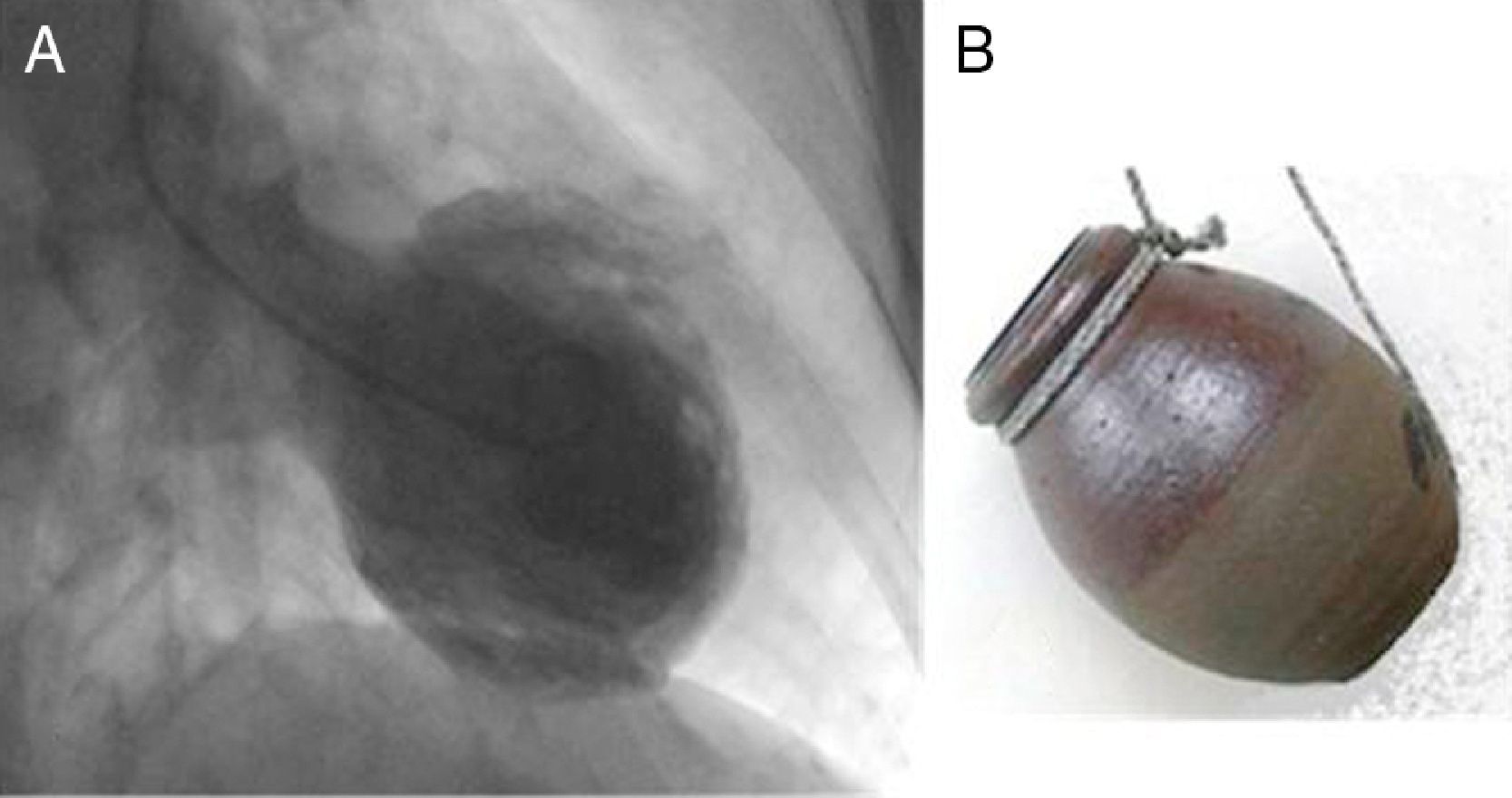
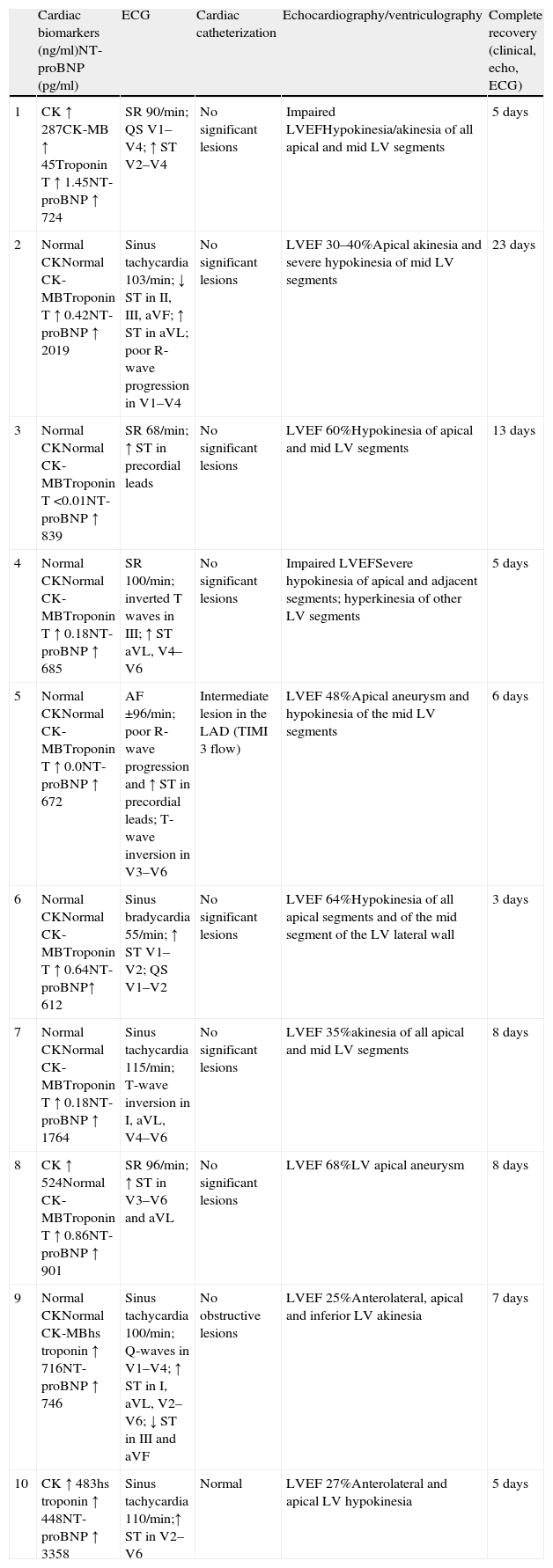
It was first described in Japan in 1990 by Sato et al., who named the syndrome after the traditional Japanese pot (tsubo) for catching octopus (tako), due to the latter's resemblance to the shape of the LV in systole (Figure 1).1
Since then, a growing number of cases have been published in different countries including Japan,2 Belgium3 and the USA,4 and, in parallel with this expansion, other terms have been coined based on cardiac morphology and clinical presentation, including transient left ventricular akinesia/dyskinesia, stress-induced cardiomyopathy, apical ballooning syndrome and broken heart syndrome.5–9
In the absence of definitive diagnostic criteria, a diagnosis of TC is made when the four modified Mayo Clinic criteria are present: (1) transient hypokinesia, dyskinesia or akinesia of the left ventricular mid segments with or without apical involvement, the wall motion abnormalities extending beyond a single epicardial vascular distribution; (2) absence of obstructive coronary disease or angiographic evidence of acute plaque rupture; (3) new electrocardiographic abnormalities (either ST-segment elevation or T-wave inversion) or modest elevation in cardiac troponin; (4) absence of pheochromocytoma or myocarditis.5
We present a series of ten cases, with the aim of comparing the characteristics of these patients with those described in the latest scientific reviews.
Case reportsIn this article, we present a series of ten cases of TC diagnosed at Hospital Garcia de Orta between January 1, 2007 and March 31, 2011, out of a total of 681 patients admitted to the cardiology department with suspected ACS.
TC is a rare acquired cardiomyopathy, as shown by the small number of reported cases, highlighting the need for a high level of clinical suspicion for its diagnosis.
Clinical, laboratory, electrocardiographic and imaging characteristics, complications, therapeutic regimens and follow-up of these patients were analyzed and are summarized in Tables 1 and 2.
Characteristics of the study population.
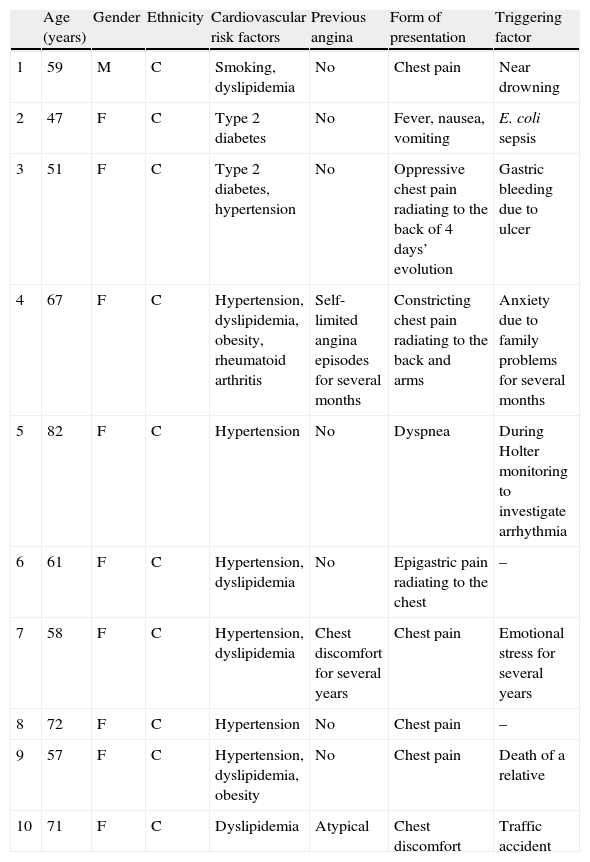
| Age (years) | Gender | Ethnicity | Cardiovascular risk factors | Previous angina | Form of presentation | Triggering factor | |
| 1 | 59 | M | C | Smoking, dyslipidemia | No | Chest pain | Near drowning |
| 2 | 47 | F | C | Type 2 diabetes | No | Fever, nausea, vomiting | E. coli sepsis |
| 3 | 51 | F | C | Type 2 diabetes, hypertension | No | Oppressive chest pain radiating to the back of 4 days’ evolution | Gastric bleeding due to ulcer |
| 4 | 67 | F | C | Hypertension, dyslipidemia, obesity, rheumatoid arthritis | Self-limited angina episodes for several months | Constricting chest pain radiating to the back and arms | Anxiety due to family problems for several months |
| 5 | 82 | F | C | Hypertension | No | Dyspnea | During Holter monitoring to investigate arrhythmia |
| 6 | 61 | F | C | Hypertension, dyslipidemia | No | Epigastric pain radiating to the chest | – |
| 7 | 58 | F | C | Hypertension, dyslipidemia | Chest discomfort for several years | Chest pain | Emotional stress for several years |
| 8 | 72 | F | C | Hypertension | No | Chest pain | – |
| 9 | 57 | F | C | Hypertension, dyslipidemia, obesity | No | Chest pain | Death of a relative |
| 10 | 71 | F | C | Dyslipidemia | Atypical | Chest discomfort | Traffic accident |
C: Caucasian.
Results of diagnostic exams and follow-up.
| Cardiac biomarkers (ng/ml)NT-proBNP (pg/ml) | ECG | Cardiac catheterization | Echocardiography/ventriculography | Complete recovery (clinical, echo, ECG) | |
| 1 | CK ↑ 287CK-MB ↑ 45Troponin T ↑ 1.45NT-proBNP ↑ 724 | SR 90/min; QS V1–V4; ↑ ST V2–V4 | No significant lesions | Impaired LVEFHypokinesia/akinesia of all apical and mid LV segments | 5 days |
| 2 | Normal CKNormal CK-MBTroponin T ↑ 0.42NT-proBNP ↑ 2019 | Sinus tachycardia 103/min; ↓ ST in II, III, aVF; ↑ ST in aVL; poor R-wave progression in V1–V4 | No significant lesions | LVEF 30–40%Apical akinesia and severe hypokinesia of mid LV segments | 23 days |
| 3 | Normal CKNormal CK-MBTroponin T <0.01NT-proBNP ↑ 839 | SR 68/min; ↑ ST in precordial leads | No significant lesions | LVEF 60%Hypokinesia of apical and mid LV segments | 13 days |
| 4 | Normal CKNormal CK-MBTroponin T ↑ 0.18NT-proBNP ↑ 685 | SR 100/min; inverted T waves in III; ↑ ST aVL, V4–V6 | No significant lesions | Impaired LVEFSevere hypokinesia of apical and adjacent segments; hyperkinesia of other LV segments | 5 days |
| 5 | Normal CKNormal CK-MBTroponin T ↑ 0.0NT-proBNP ↑ 672 | AF ±96/min; poor R-wave progression and ↑ ST in precordial leads; T-wave inversion in V3–V6 | Intermediate lesion in the LAD (TIMI 3 flow) | LVEF 48%Apical aneurysm and hypokinesia of the mid LV segments | 6 days |
| 6 | Normal CKNormal CK-MBTroponin T ↑ 0.64NT-proBNP↑ 612 | Sinus bradycardia 55/min; ↑ ST V1–V2; QS V1–V2 | No significant lesions | LVEF 64%Hypokinesia of all apical segments and of the mid segment of the LV lateral wall | 3 days |
| 7 | Normal CKNormal CK-MBTroponin T ↑ 0.18NT-proBNP ↑ 1764 | Sinus tachycardia 115/min; T-wave inversion in I, aVL, V4–V6 | No significant lesions | LVEF 35%akinesia of all apical and mid LV segments | 8 days |
| 8 | CK ↑ 524Normal CK-MBTroponin T ↑ 0.86NT-proBNP ↑ 901 | SR 96/min; ↑ ST in V3–V6 and aVL | No significant lesions | LVEF 68%LV apical aneurysm | 8 days |
| 9 | Normal CKNormal CK-MBhs troponin ↑ 716NT-proBNP ↑ 746 | Sinus tachycardia 100/min; Q-waves in V1–V4; ↑ ST in I, aVL, V2–V6; ↓ ST in III and aVF | No obstructive lesions | LVEF 25%Anterolateral, apical and inferior LV akinesia | 7 days |
| 10 | CK ↑ 483hs troponin ↑ 448NT-proBNP ↑ 3358 | Sinus tachycardia 110/min;↑ ST in V2–V6 | Normal | LVEF 27%Anterolateral and apical LV hypokinesia | 5 days |
AF: atrial fibrillation; Echo: echocardiographic; hs: high-sensitivity; LV: left ventricular; LVEF: left ventricular ejection fraction; SR: sinus rhythm.
There was a clear predominance of females (9:1); ages ranged between 47 and 82 years (mean 62). All patients were Caucasian, and so no ethnic trend could be assessed.
Cardiovascular risk factors identified included, in descending order, hypertension (70%), dyslipidemia (60%), type 2 diabetes (20%), obesity (20%) and smoking (10%). One patient had a previous diagnosis of rheumatoid arthritis, which is considered an independent risk factor for coronary artery disease (CAD) with mandatory screening. All patients had at least one cardiovascular risk factor (40% had one, 40% two and 20% three). The presence of these risk factors complicates differential diagnosis with myocardial infarction (MI), since they mean that concomitant CAD is more likely.
The most common form of presentation was chest pain (80%), followed by dyspnea (10%) and, less specifically, fever syndrome (10%). Only two patients reported previous chest discomfort, which had been episodic, self-limited, long-standing (months or years) and in all cases associated with anxiety.
There was a wide range of triggering stress factors, emotional (near drowning, family problems, Holter monitoring, death of a relative, traffic accident), or physical (sepsis, gastric bleeding). In two cases no trigger was identified.
The results of diagnostic exams and follow-up are summarized in Table 2.
Levels of cardiac biomarkers (creatine kinase [CK] and troponin) and results of the electrocardiogram (ECG), echocardiography, ventriculography and cardiac catheterization were analyzed. Troponin levels were elevated in 90% of patients but CK was elevated in only 30%. There was a marked discrepancy between the modest elevation in cardiac enzymes and LV wall motion abnormalities. N-terminal brain natriuretic peptide (NT-proBNP) was elevated in all cases.
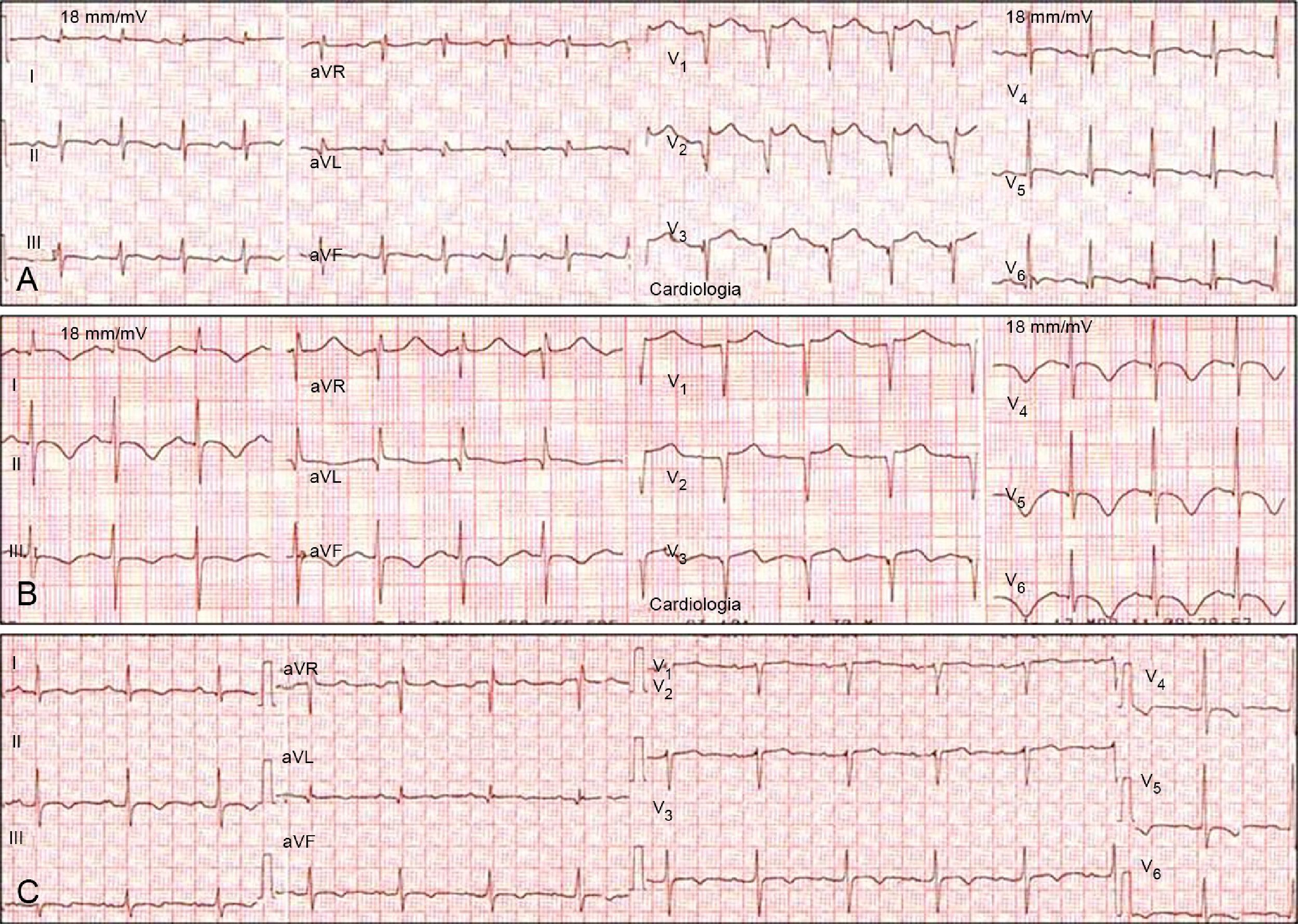
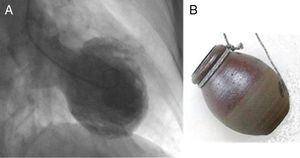
The most frequent ECG abnormalities were ST-segment elevation in the precordial leads (80%) mimicking anterior MI (Figure 2). Other alterations were T-wave inversion (30%), ST-segment depression in the inferior leads (20%) and poor R-wave progression in the precordial leads (20%).
Serial electrocardiograms in a patient with takotsubo cardiomyopathy mimicking anterior myocardial infarction. (A) Acute phase: sinus tachycardia and ST-segment elevation in V2–V6; (B) subacute phase (2nd day): T-wave inversion in I, II, III, aVF, and V3–V6; (C) subacute phase (5th day): beginning of normalization and reduction in T-wave amplitude.
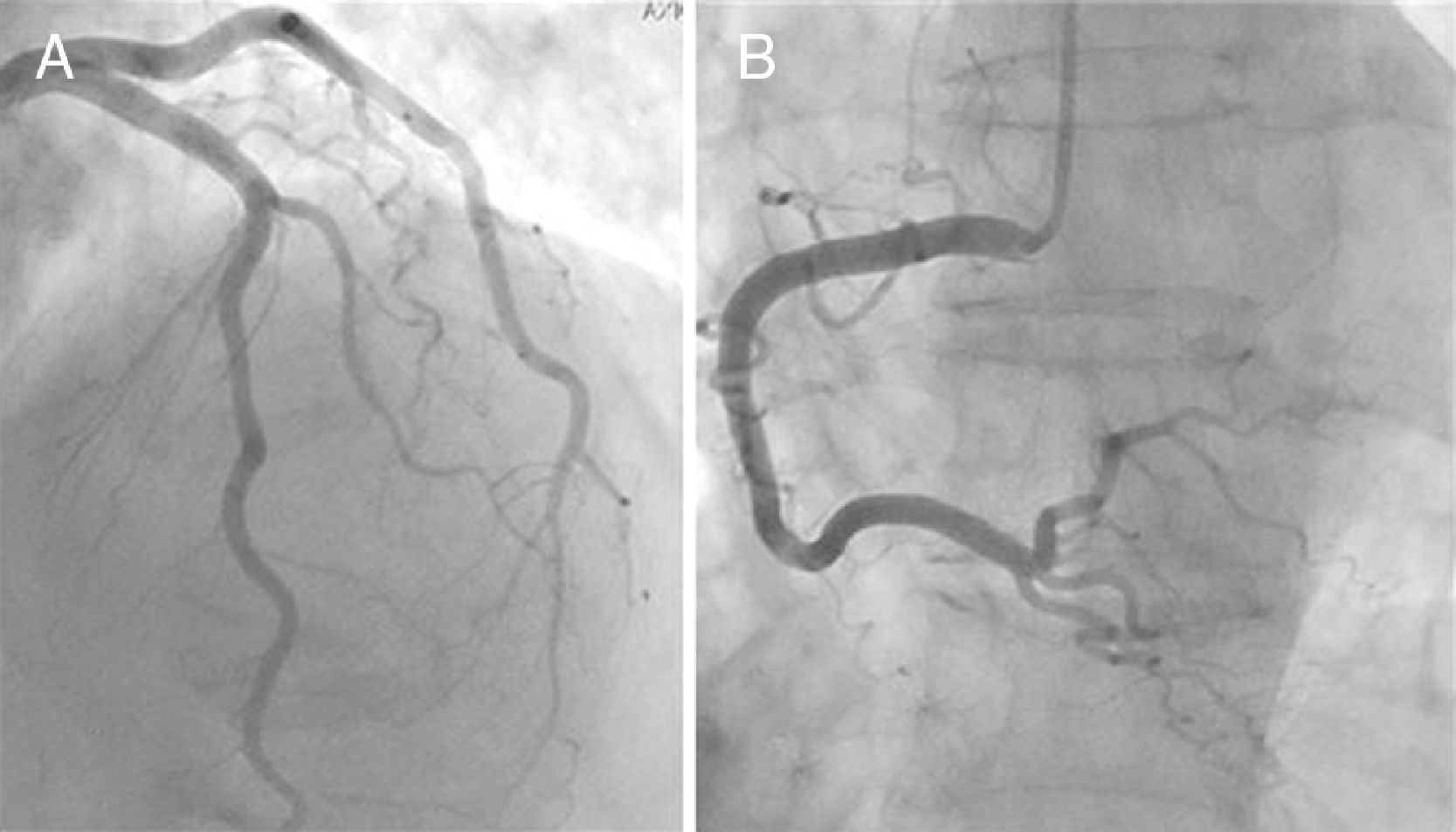
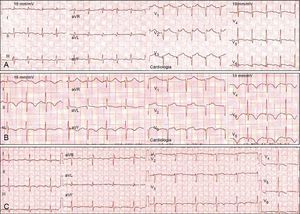
Echocardiography and ventriculography revealed akinesia/hypokinesia in all cases, in the apical (100%) and/or mid segments (90%), with impaired left ventricular ejection fraction (LVEF), ranging between 25% and 68% (Figure 1). Cardiac catheterization identified an intermediate lesion in the left anterior descending artery in one patient but no significant lesions in the others (Figure 3).
All patients in this series had a good prognosis, which is typical of the reversible nature of TC, but this does not detract from the seriousness of the acute phase, in which LV systolic dysfunction can be severe. There was one case of cardiogenic shock.
Therapy was based on cardiac support, with angiotensin-converting enzyme inhibitors and beta-blockers, which are recognized as important for LV remodeling and recovery of cardiac function; risk factor control with aspirin, statins and oral antidiabetic agents; and ventilatory, inotropic and vasoactive support in the patient with cardiogenic shock.
In parallel with clinical improvement, normalization of cardiac biomarkers and echocardiographic and ECG alterations was seen in all patients during hospital stay, within a mean of eight days. In subsequent outpatient follow-up consultations they were all asymptomatic, with normal exams, which is typical of the good prognosis due to the completely reversible nature of TC (Table 2).
DiscussionThe real prevalence of TC is unknown5–9; however, recent statistical analyses have indicated a prevalence of 1.7–2.2% in cases of suspected ACS.5–7
A review of the literature indicates that it is more common in individuals aged between 62 and 75 years (mean 67),6 and in women (90%), particularly postmenopausal.5–8
TC is usually associated with emotional or physical stress, including death of a relative, diagnosis of a serious disease, natural disasters, financial problems, traffic accidents, worsening of a chronic illness, surgery, admission to an intensive care unit, or use of illicit drugs.5–8 Cases have been reported following the use of phenylephrine.6 Patients with TC have been shown to have elevated catecholamine levels.6 In some cases no stress trigger has been identified.7
TC is a differential diagnosis with ACS. Clinically, the most common forms of presentation are chest pain (67%) and dyspnea (17%), although more serious settings, such as cardiogenic shock (4.2%) and ventricular fibrillation (1.5%), have been reported.7
The ECG abnormalities most often observed in the acute and subacute phases are ST-segment elevation (67–75%) and T-wave inversion (61%). The ST-segment elevation involves the precordial leads V2–V3 in 90% of cases. New-onset left or right bundle branch block and (usually transient) pathological Q waves have also been reported (6–31%). However, some 15% of TC patients have normal ECG or non-specific alterations.6 In a retrospective study of 33 patients with TC, Kosuge et al. proposed criteria with high specificity and sensitivity to distinguish TC from anterior MI: absence of abnormal Q waves, absence of reciprocal changes, presence of ST-segment depression in aVR, and absence of ST-segment elevation in V1.10 This differential diagnosis is particularly important given the differences in treatment for TC versus ST-segment elevation MI (STEMI), especially in hospitals where thrombolysis is the only therapeutic option, since the timing of this therapy for STEMI is critical and will be affected by a clinical suspicion of TC. However, given the much higher prevalence of STEMI compared to TC, unconfirmed clinical suspicion is not sufficient reason not to perform thrombolysis.5
Cardiac biomarkers, specifically troponin I and T, are elevated in 90% of cases, although to a lesser extent than in STEMI.5–8 Peak troponin T of 0.64 ng/ml and peak troponin I of 8.6 ng/ml have been reported6; CK may also be elevated. NT-proBNP levels are usually raised, particularly in patients who develop severe LF systolic dysfunction.5–8
Echocardiography and left ventriculography reveal specific alterations: akinesia/dyskinesia of the LV mid and apical segments, with an area of akinesia corresponding to the territory perfused by more than one coronary artery. In a few cases, hypokinesia is limited to the mid segments, with no apical involvement; this is known as atypical TC.11 LVEF is 20–49%.6
Diagnosis is confirmed by cardiac catheterization showing absence of or non-obstructive CAD.
These characteristics are the key to differentiating TC from other reversible conditions that have similar clinical features and ST-segment elevation, including cardiac syndrome X, Prinzmetal angina, myocarditis and cocaine use.5
The etiology of TC remains unknown. Various mechanisms have been put forward to explain its acute, reversible and partly environmentally influenced nature and are still under investigation: coronary spasm, microvascular ischemia, catecholamine cardiotoxicity, isolated plaque rupture in the left anterior descending artery, and acute dynamic LV outflow tract obstruction.8,9 The most widely accepted theory at present is of excessive adrenergic stimulation, associated with the release of large quantities of catecholamines, probably in response to internal or external stress, producing the characteristic clinical, laboratory and imaging features.5–9
Prognosis is generally favorable, with complete recovery of all the characteristic alterations, usually within two to four weeks.5,6 The reversibility of the condition and the fact that its pathophysiology is unclear mean that there is no specific treatment; supportive therapy is recommended, according to the degree of systolic dysfunction, and directed at acute complications, which occur in 20% of cases and can include cardiogenic shock, acute pulmonary edema, arrhythmias, LV thrombi leading to embolism and even death.6,9
ConclusionTC is an acquired cardiomyopathy that is increasingly recognized worldwide. It is considered rare, but its low prevalence probably masks a large number of undiagnosed cases, due to its similarity to ACS.
It is thus essential to take account of certain clues to the diagnosis that have been reported and refined over the years in published case series and that serve to alert the physician to the existence of this entity. The main ones are age, gender, presence of emotional or physical stress, and the discrepancy between the modest rise in cardiac enzymes and significant LV wall motion abnormalities.
To summarize, a diagnosis of TC requires a high index of clinical suspicion and the timely use of diagnostic exams other than ECG and measurement of cardiac biomarkers, of which the most crucial are echocardiography (which demonstrates LV wall motion abnormalities) and cardiac catheterization (to confirm the absence of significant coronary lesions).
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Cesário V, et al. Miocardiopatia de takotsubo num serviço de cardiologia. Rev Port Cardiol. 2012. http://dx.doi.org/10.1016/j.repc.2012.01.018.