Heart failure (HF) is one of the most prevalent conditions worldwide and despite therapeutic advances, its prognosis remains poor. Among the multiple comorbidities in HF, sleep-disordered breathing (SDB) is frequent and worsens the prognosis. Preliminary observational studies suggested that treatment of SDB could modify the prognosis of HF, and the issue has gained importance in recent years. The diagnosis of SDB is expensive, slow and suboptimal, and there is thus a need for screening devices that are easier to use and validated in this population. The first-line treatment involves optimization of medical therapy for heart failure. Continuous positive airway pressure (CPAP) is used in patients who mainly suffer from obstructive sleep apnea. In patients with predominantly central sleep apnea, CPAP is not sufficient and adaptive servo-ventilation (ASV), despite promising results in observational studies, showed no benefit in patients with symptomatic HF and reduced ejection fraction in the SERVE-HF randomized trial; on the contrary, there was unexpectedly increased mortality in the ASV group compared to controls, and so ASV is contraindicated in these patients, calling into question the definition and pathogenesis of SDB and risk stratification in these patients. There are many gaps in the evidence, and so further research is needed to better understand this issue: definitions, simple screening methods, and whether and how to treat SDB in patients with HF.
A insuficiência cardíaca (IC) é uma das entidades mais prevalentes em todo o mundo e o seu prognóstico, apesar dos avanços terapêuticos, continua reservado. De entre as múltiplas comorbilidades, os distúrbios respiratórios do sono (DRS) são frequentes na IC e agravam o seu prognóstico. Estudos preliminares observacionais sugeriam que o tratamento dos DRS poderia modificar o prognóstico da IC, motivo pelo qual o tema tem ganho importância nos últimos anos. O diagnóstico dos DRS é caro e moroso, pelo que subótimo, com necessidade de dispositivos de rastreio mais fáceis de usar e validados nesta população de doentes. O tratamento em primeira linha passa pela otimização da terapêutica médica da IC. A pressão positiva contínua na via aérea (CPAP) é utilizada em doentes com predomínio de apneia obstrutiva. Já nos doentes com predomínio de apneia central, o CPAP não é suficiente e a servo-ventilação adaptativa (SVA), apesar de ter mostrado resultados promissores em estudos observacionais, não mostrou benefício nos doentes com IC sintomática e fração de ejeção deprimida no estudo aleatorizado SERVE-HF; pelo contrário, registou-se, inesperadamente, um aumento da mortalidade no grupo do SVA em relação ao grupo controlo, razão pela qual está contraindicada nestes doentes, pondo em causa a definição e etiopatogenia dos DRS e estratificação de risco destes doentes. Existem muitas lacunas na evidência, pelo que será necessária mais investigação para um melhor entendimento deste tema: definição, métodos simples de rastreio, bem como da necessidade ou não, e melhor forma de tratar os DRS nos doentes com IC.
Heart failure (HF) is recognized as a worldwide epidemic, with high morbidity and mortality. Its prevalence is around 1-2% and can exceed 10% in those aged 70 years or over.1 In Portugal, it ranges from 1.36% in those aged over 25 years to 16% in those over 80; the prevalence of HF with reduced ejection fraction (HFrEF) and HF with preserved ejection fraction (HFpEF) is 1.3% and 1.7%, respectively.2 Despite therapeutic advances, the short- and long-term prognosis of HF remains poor, which has prompted an ongoing search for risk modifiers and treatments that can demonstrably improve prognosis. In this context, in recent years there has been considerable interest in sleep-disordered breathing (SDB), a condition that is frequently associated with worse prognosis in HF patients. The main form of treatment for SDB in HF is non-invasive ventilation, either by continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BPAP) for patients who mainly suffer from obstructive sleep apnea (OSA), or by adaptive servo-ventilation (ASV) for those with predominantly central sleep apnea (CSA). It has been hypothesized that in addition to optimal HF therapy, such treatment could bring added benefits for HF patients.
Preliminary observational studies suggested that treatment of SDB could modify the prognosis of HF, reducing hospitalizations and long-term mortality.3 However, these expectations were frustrated by the recently published results of the SERVE-HF trial,4 in which treatment of SDB by ASV in fact increased mortality in patients with symptomatic HFrEF. The discrepancy between the results of observational studies and those of SERVE-HF call into question the pathogenesis of SDB in HF, challenging clinicians and researchers to improve their knowledge in this area. To this end, we present a review of the subject.
ConceptsSleep apnea syndrome is characterized by repeated pauses in breathing resulting in sleep fragmentation, daytime sleepiness and blood oxygen desaturation. According to the American Academy of Sleep Medicine, apnea is defined as a drop of ≥90% in respiratory flow from baseline lasting for ≥10 s, and hypopnea as a drop of ≥30% in respiratory flow from baseline lasting for ≥10 s in association with either ≥3% arterial oxygen desaturation or an arousal.5
Patients with HF can suffer from CSA, OSA, or both. The prevalence of CSA increases with increasing severity of HF. OSA is caused by upper airway collapse with increased reflex respiratory effort. Predisposing factors include obesity and unfavorable craniofacial structure.
In CSA, there is no strengthening of the respiratory reflex during episodes of apnea or hypopnea. The underlying mechanism is increased sensitivity of chemoreceptors in the brainstem and the carotid bodies to small variations in partial pressure of carbon dioxide (PaCO2), leading to inappropriate hyperventilation.6,7 The resulting reduction in PaCO2 weakens the respiratory stimulus and apnea or hypopnea occurs; when PaCO2 rises, the cycle repeats. In addition to this excessive respiratory response to CO2 levels, another mechanism at play in hyperventilation is stimulation of vagal mechanoreceptors (J receptors) due to pulmonary congestion via vagal afferents.8–11 CSA with a cyclic pattern of hyperventilation-apnea-hyperventilation is known as Cheyne-Stokes respiration.
The apnea-hypopnea index (AHI) is the number of events (apnea and/or hypopnea) per hour of sleep. To be considered as CSA, the number of central events must be more than 50% of the total number of events recorded by polysomnography. Sleep apnea can be classified as mild (AHI 6-14/h), moderate (15-29/h) or severe (>30/h). Although this is not universally accepted, there is general agreement that the threshold requiring treatment in symptomatic patients is an AHI >15/h.12 This cutoff is further complicated in HF patients by the fact that the clinical features of SDB in HF are atypical, and that the symptoms of HF can mimic those of SDB and vice versa.
PathophysiologyOSA is considered an independent risk factor for HF.13 The main pathophysiological mechanisms by which it worsens HF are increased intrathoracic pressure, resulting in raised pre- and afterload leading to left ventricular compromise, excessive activation of the sympathetic nervous system, and hypoxemia.14
CSA is thought to be a consequence rather than a cause of HF.15 Besides hypoxemia, sympathetic activation is predominant, resulting in increased peripheral vascular resistance, tachycardia, and stimulation of the renin-angiotensin-aldosterone system.
In both forms, variations in blood pressure and heart rate contribute to endothelial dysfunction and elevated inflammatory markers, particularly C-reactive protein.16,17
Patients with SDB are at increased risk of ventricular and other arrhythmias, especially atrial fibrillation.18–20 In an observational study, appropriate therapies from an implantable cardioverter-defibrillator were more frequent at night in patients with OSA and during the day in those with CSA.21 In addition to arrhythmic risk, SDB also is associated with increased risk of atherothrombotic phenomena22 and metabolic alterations, particularly type 2 diabetes.23
Recently, and particularly after the SERVE-HF trial,4 the idea has gained ground that CSA in HF patients is a compensatory mechanism that has some beneficial effects, including the provision of periodic rest to fatigue-prone respiratory muscles, attenuation of excessive sympathetic nervous activity, avoidance of hypercapnic acidosis and increases in end-expiratory lung volume and intrinsic positive airway pressure.24
EpidemiologyThe prevalence of SDB in HF is high (over 50%). In a study of 700 patients with HFrEF in New York Heart Association (NYHA) class ≥II, Oldenburg et al. found a prevalence of 76% (40% CSA and 36% OSA) using a cutoff AHI value of ≥5/h.25 Schulz et al., studying 203 patients with HFrEF in NYHA class II-III, observed a prevalence of 71% (43% CSA and 28% OSA) with a cutoff of AHI ≥10/h,26 while in a study by Vazir et al. of 55 men with mildly symptomatic HFrEF, 53% presented SDB defined as AHI ≥15/h.27
In patients with HFpEF, the prevalence of SDB is also high. Using a cutoff of AHI >10/h, Chan et al. found a prevalence of 56%, mainly OSA,28 while Bitter et al., using a definition of AHI ≥15/h, observed a prevalence of 69%, mainly OSA.29
A prospective observational study in Germany including 1500 patients with HFrEF revealed a prevalence of SDB of 80% despite optimal HF therapy.30 Snoring was more prevalent in patients with OSA, while atrial fibrillation and ischemic cardiomyopathy were more common in the CSA group. The most frequent symptoms were nocturia, witnessed apnea and fatigue, which illustrates the difficulty of distinguishing these symptoms from those of HF itself.
DiagnosisIn daily clinical practice, HF patients with SDB rarely report daytime sleepiness, possibly because of sympathetic hyperactivation, and thus many of them remain undiagnosed.31 Questionnaires such as the Epworth Sleepiness Scale,32 the Berlin Questionnaire33 and the Maislin Questionnaire34 are used to screen for SDB, although their usefulness has been questioned.
Polysomnography remains the gold standard for diagnosis of SDB. However, despite the comprehensive and detailed information it provides, polysomnography requires specialized technical and human resources, and is slow and expensive, with long waiting lists. There is thus a need for simpler and more accessible protocols, and a large number of portable devices have been developed for this purpose using techniques that include simplified cardiorespiratory polygraphy with single or multiple channels, oximetry and/or nasal air flow,35,36 which could be alternatives to polysomnography for screening purposes.
One such device is the ApneaLink (ResMed Corp, San Diego, CA). A study by Erman et al. validating this device in screening for SDB in 59 diabetic patients showed that the ApneaLink provided reliable information and was easy to use, with high sensitivity and specificity in calculating AHI compared to polysomnography, especially for AHI ≥15/h (sensitivity 91% and specificity 95%).37 Ragette et al. also found an excellent correlation between the ApneaLink and polysomnography, with better results for AHI ≥10/h.38 In addition, the device showed good diagnostic ability compared to polysomnography (sensitivity 87.1% and specificity 94.9%) for detecting CSA events in a study of 70 individuals (41 with HF) with Cheyne-Stokes respiration.39
PrognosisGiven its pathophysiology, it is no surprise that SDB is associated with worse prognosis in HF patients. OSA is described as a cardiovascular risk marker that is more often found in mild forms of HF (NYHA I-II), while CSA tends to be an indicator of more severe cases (NYHA III-IV).
OSA was associated with increased mortality in patients with HFrEF over a mean follow-up of three years (hazard ratio [HR] 2.81, p=0.029)40 and in patients with ischemic HF after a mean follow-up of 32 months (HR 3.03, p=0.043).41 Other authors report similar results in patients in whom CSA was identified during hospital stay, with lower survival in those with more severe disease.42
In a study including 784 patients with systolic HF, CSA was an independent predictor of six-month cardiac readmission,43 and in another study, the same authors reported that both OSA and CSA are independent risk factors for long-term post-discharge mortality in patients hospitalized for acute HF.3
In patients with HF and CSA, one of the most important prognostic indicators is the nocturnal hypoxemic burden. In a study of 963 patients with HFrEF, time with peripheral oxygen saturation <90% (T90) was associated with a 16% increase in all-cause mortality per hour of T90 in multivariate analysis, while AHI was no longer significant after adjustment for confounding factors.44 The best cutoff for mortality in this population was a T90 of at least 22 min. According to these authors, nocturnal hypoxemic burden could be an important therapeutic target and reducing hypoxemia could be more useful than reducing AHI.
TreatmentObstructive sleep apneaPositive pressure devices are an established therapy in the treatment of OSA in patients without HF.45 In OSA, nocturnal CPAP reduces sleepiness, improves quality of life and left ventricular ejection fraction (LVEF), and lowers catecholamine levels.46,47 In an observational study of OSA in HF, CPAP was associated with lower death and hospitalization rates (Table 1).48
Studies on obstructive sleep apnea in heart failure.
| Authors | Design | Main conclusions |
|---|---|---|
| Mansfield et al.47 | Randomized trial in 40 patients (CPAP vs. placebo) | Improvements in ventricular function, quality of life, and sympathetic nervous system activity with CPAP |
| Kasai et al.48 | Observational study of 88 patients | Reduced risk of death and hospitalization for HF with CPAP |
| Mehra et al.49 | Systematic review and meta-analysis of 14 observational studies | Reduced nocturnal oxygen desaturation |
| Bucca et al.50 | Observational study of 15 patients | Reduced AHI with diuretics |
| Araghi et al.51 | Systematic review and meta-analysis of 7 randomized trials | Reduced AHI with reduction in body weight |
AHI: apnea-hypopnea index; CPAP: continuous positive airway pressure; HF: heart failure.
In patients who do not tolerate CPAP, nocturnal oxygen therapy reduces desaturation, although with prolonged apnea and hypopnea episodes.49
Optimization of HF therapy improves cardiac output and reduces peripheral edema, and diuretics improve OSA in patients with HF and hypervolemia.51 Weight loss also significantly reduces AHI, according to a meta-analysis of seven randomized trials (Table 1).51
Central sleep apneaIndications for treatment and the choice of treatment modality for CSA in HF patients are the subject of debate. According to the latest guidelines, the first step is to optimize HF therapy.52 Anti-remodeling therapy with angiotensin-converting enzyme inhibitors and adrenergic blockers reduces CSA in congestive HF.53,54 In a randomized trial,55 theophylline, which stimulates respiration by competing with adenosine, reduced CSA in 50% of patients, but due to its narrow therapeutic window it is not used in clinical practice. Acetazolamide also reduces CSA and daytime sleepiness and improves sleep quality in HF patients, by promoting diuresis and metabolic acidosis, which stimulates ventilation.56 However, acetazolamide may cause hypokalemia, with pro-arrhythmic effects. According to a recent meta-analysis, cardiac resynchronization (CRT) in patients with CSA, HFrEF and complete left bundle branch block led to a mean reduction in AHI of 13/h, although this improvement was not seen in patients with OSA (Table 2).57 In an observational study, atrial overdrive pacing resulted in an additional reduction in CSA when combined with CRT.58
Studies on central sleep apnea in heart failure.
| Authors | Design | Main conclusions |
|---|---|---|
| Walsh et al.53 and Tamura et al.54 | Observational studies | Improved AHI under captopril and carvedilol |
| Javaheri et al.55 | Randomized trial with 15 patients (theophylline vs. control) | Fewer apnea and hypopnea episodes with theophylline |
| Javaheri et al.56 | Randomized trial with 12 patients (acetazolamide-KCl and control) | Improvement in CSA and symptoms with acetazolamide |
| Lamba et al.57 | Systematic review and meta-analysis of 9 observational studies | Reduced AHI with CRT (no benefit in patients with OSA) |
| Nakao et al. (CHF-HOT)65 | Post-hoc analysis of 2 randomized trials with 97 patients (nocturnal oxygen vs. control) | Improved CSA, quality of life and ventricular function with nocturnal oxygen |
| Bradley et al. (CANCAP)67 | Randomized trial with 258 patients with reduced LVEF (CPAP vs. control) | No significant differences in primary endpoint of number of hospitalizations, mortality or transplantation |
| Köhnlein et al.70 | Randomized trial with 35 patients with reduced LVEF (CPAP vs. BPAP – crossover) | Improvement in Cheyne-Stokes respiration with CPAP and BPAP |
| Sharma et al.72 | Systematic review and meta-analysis of 14 observational studies with 538 patients (ASV vs. control) | Improved AHI, LVEF and functional capacity with ASV |
| Cowie et al. (SERVE-HF)4 | Randomized trial with 1325 patients with CSA and reduced LVEF (ASV vs. control) | No differences in primary endpoint (HR 1.13; p=0.10). Increased overall and cardiovascular mortality in patients treated by ASV |
| Abraham et al.76 | Observational study with 57 patients with CSA (phrenic nerve stimulation vs. control) | 55% reduction in AHI with phrenic nerve stimulation |
AHI: apnea-hypopnea index; ASV: adaptive servo-ventilation; BPAP: bilevel positive airway pressure; CPAP: continuous positive airway pressure; CRT: cardiac resynchronization; CSA: central sleep apnea; HR: hazard ratio; LVEF: left ventricular ejection fraction; OSA: obstructive sleep apnea.
Implantation of a left ventricular assist device was associated with improvement in CSA and exercise capacity in HF patients.59 Cardiac transplantation60 and mitral valve replacement, when indicated,61 have also been followed by improvement in Cheyne-Stokes breathing.
Nocturnal oxygen therapy reduces CSA and sympathetic activity, improving quality of life and exercise capacity in HF.62–64 A post-hoc analysis of the CHF-HOT randomized trials including 97 patients with HF and CSA showed reduced AHI and improved LVEF and functional class with nocturnal oxygen therapy compared to medical therapy alone (Table 2).65
Positive pressure devices reduce pulmonary edema by decreasing ventricular pre- and afterload and hence CSA. Small studies, most of them observational, in patients with HF and CSA have shown that CPAP also improves AHI and LVEF and reduces sympathetic activity, with a tendency for lower mortality.66 A randomized trial, the Canadian Positive Airway Pressure for Patients with Central Sleep Apnea and Heart Failure Trial (CANCAP), compared CPAP with optimal medical therapy in 258 patients with HFrEF and CSA. After a mean follow-up of two years, no significant differences were found in the primary outcome of death from all causes and heart transplantation (Table 2).67 It should be noted that CPAP had only a weak effect on CSA in this trial, reducing mean AHI to 19/h, which was more than the 15/h inclusion criterion. A post-hoc analysis of this trial showed that prognosis was better for patients in whom CPAP resulted in CSA being reduced to less than 15 events/h.68
An observational study involving 296 patients with chronic HFrEF and sleep apnea (central and obstructive) revealed lower long-term mortality in patients with severe apnea (AHI >22.5) in those treated with CPAP compared to untreated patients.69
BPAP has been described as a valid alternative for patients who do not tolerate CPAP and as equally effective in improving NYHA functional class and sleep quality in HF patients with CSA (Table 2).70
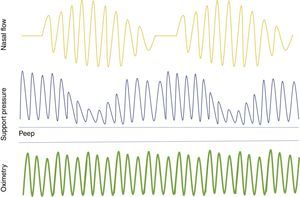
ASV is a positive pressure technology that controls airway pressure according to the patient's ventilation, re-establishing a normal breathing pattern. It is a promising alternative to CPAP for the treatment of CSA in HF (Figure 1).71 A meta-analysis of studies on CSA and HF showed that ASV reduced AHI and improved cardiac function and exercise capacity,72 while another meta-analysis demonstrated a significant reduction in all-cause mortality with ASV compared to CPAP in patients with HF and SDB.73
Adaptive servo-ventilation keeps the airway open by means of positive end-expiratory pressure and provides inspiratory pressure support during spontaneous breathing. When apnea occurs, it increases support pressure to provide minimum ventilatory reserve. As soon as spontaneous breathing returns, support is reduced. PEEP: positive end-expiratory pressure.
In view of these apparent benefits of ASV, the SERVE-HF trial randomized 1325 patients with HFrEF and CSA to guideline-based medical treatment (controls) or ASV for at least five hours per night, seven days per week.4 After a median follow-up of 31 months, despite effective control of CSA by ASV, there were no differences in the primary study endpoint of death from any cause, lifesaving cardiovascular intervention, or unplanned hospitalization for worsening heart failure. The event rate for the primary endpoint was 54.1% in the ASV group and 50.8% in the control group (HR 1.13; p=0.10). Furthermore, the addition of ASV to optimal medical treatment had no beneficial effect on quality of life, six-minute walk test or symptoms. On the contrary, there was higher all-cause and cardiovascular mortality in the ASV group than in controls (34.8% vs. 29.3%, HR 1.28; p=0.01 and 29.9% vs. 24.0%, HR 1.34; p=0.006). The early and sustained increase in cardiovascular mortality with ASV was unexpected in light of the results of previous meta-analyses and there has been considerable speculation concerning the causes of these results and alternative treatments (Table 2). One possible explanation is that in some HF patients CSA could in fact be a compensatory mechanism that provides periods of rest to respiratory muscles and reduces excessive sympathetic nervous activity. Another theory is that positive airway pressure may reduce systolic ejection volume and hence cardiac output, especially in the presence of low pulmonary wedge pressure. A recent sub-analysis of the SERVE-HF trial examined the types of death that contributed to the higher mortality in the ASV group, and found that patients in this group without previous hospital admission for worsening heart failure were at higher risk for cardiovascular death, suggesting that the additional mortality was due to sudden death. This is supported by the fact that it was patients with the lowest LVEF (<30%) and without implantable cardioverter-defibrillators who were most affected (HR 5.2; p=0.026).74
An interesting finding of SERVE-HF is that although ASV reduced AHI and the oxygen desaturation index, mean T90 was only decreased from 51 to 19 min, and so many patients still had a considerable hypoxemic burden.
The search for explanations of the unexpected results of SERVE-HF has led to the idea that the therapeutic target in patients with HFrEF and SDB should be T90 rather than AHI, and has cast doubt on the usefulness of AHI for defining SDB or for risk stratification in these patients. Nevertheless, in the light of SERVE-HF, ASV is now contradicted in patients with CSA and symptomatic HF with LVEF ≤45%. It should be noted that the study population was composed exclusively of patients with HFrEF, most of whom had CSA, and therefore the results cannot be extrapolated to patients with HFpEF or predominantly OSA. A randomized trial, ADVENT-HF, is under way in patients with HFrEF and CSA or OSA (ClinicalTrials.gov identifier: NCT01128816).
Transvenous phrenic nerve stimulation has been proposed as a new, more physiological approach in the treatment of CSA that can prevent apnea, eliminating the need for positive airway pressure or patient compliance. An implanted device similar to a pacemaker is used, with an electrode in the left pericardiophrenic or right brachiocephalic vein, which stimulates the phrenic nerve when no impulse is detected for a specified period of time. An observational study of 30 patients with HF and CSA showed that unilateral transvenous phrenic nerve stimulation significantly reduced the number of CSA episodes,75 while in a prospective study of 57 patients it led to improvements in sleep, oxygenation and quality of life.76 A multicenter randomized trial, the Pivotal Trial of the remedē System (ClinicalTrials.gov identifier: NCT01816776), is under way that will evaluate the safety and effectiveness of the remedē system (Respicardia, Inc.). Although this is an invasive technique, it has attracted greater attention since the negative results of SERVE-HF.
The futureFollowing the publication of SERVE-HF, many questions remain concerning the pathophysiology of SDB in HF patients, as well as how to classify and investigate the severity of the syndrome, in particular regarding AHI and T90, and what should be the therapeutic targets. The effects on morbidity and mortality of decreasing nocturnal hypoxemia, as opposed to reducing AHI, need to be tested, as do other therapeutic modalities, particularly oxygen supplementation and phrenic nerve stimulation. Among other challenges, there is a need for a consensual definition of SDB in HF and for simple screening methods, and there has still been no unequivocal identification of the best therapeutic targets and most appropriate treatments for each phenotype of the disease (Table 3). It is clear that HF patients with SDB have a poor prognosis, but it remains to be demonstrated whether treating SDB improves quality of life and survival in these patients, and confirmation is still awaited that SDB is a risk factor and not simply an indicator of HF severity.
Areas for future research.
| An agreed definition of SDB |
| Simpler, faster and cheaper diagnostic methods |
| Establishing a consensus on T90 vs. AHI reduction as the therapeutic target |
| Role of oxygen therapy |
| Treatment of other populations with HF and SDB (HFpEF, OSA) |
| The role of ASV |
| Benefits of phrenic nerve stimulation in HFrEF and SDB |
AHI: apnea-hypopnea index; ASV: adaptive servo-ventilation; HF: heart failure; HFpEF: heart failure with preserved ejection fraction; HFrEF: heart failure with reduced ejection fraction; OSA: obstructive sleep apnea; SDB: sleep disordered breathing; T90: time with oxygen saturation <90%.
SDB is a common comorbidity in HF that worsens morbidity and mortality, and thus HF specialists, especially cardiologists and internists, must be able to suspect and diagnose the condition and refer patients to sleep specialists, as well as to monitor the effects of treatments administered to their patients.
Results of studies on various treatments have been inconsistent. Following SERVE-HF, ASV is contraindicated for patients with HFrEF and CSA. However, this finding cannot necessarily be extrapolated to other populations, such as those with HFpEF or CSA. SERVE-HF has opened new horizons for research, and studies currently under way may be crucial in defining the most appropriate approach to SDB in HF.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Carmo J, Araújo I, Marques F, et al. Distúrbios respiratórios do sono na insuficiência cardíaca: o estado da arte depois do estudo SERVE-HF. Rev Port Cardiol. 2017;36:859–867.