Aortic coarctation (AC) represents ∼7% of congenital cardiac diseases and is usually diagnosed in childhood or early adult life, depending on the severity of obstruction and associated malformations. Left untreated fewer than 20% of patients survive to age 50. We describe a case of thoracic AC, diagnosed at age 61, in a woman with known hypertension since age 45. At age 56 the patient was admitted with a subarachnoid hemorrhage and, during cerebral angiography, a thoracic aortic aneurysm was detected. Four years later the patient was referred to the outpatient hypertension clinic due to uncontrolled hypertension and cardiac failure. The echocardiogram disclosed left ventricular hypertrophy and aggressive treatment failed to control her hypertension. At age 61, due to lower limb muscular fatigue, arterial Doppler ultrasound was performed that revealed symmetrically decreased ankle/brachial pressure index, suggesting aortic stenosis. MRI angiography enabled a diagnosis of AC with a large poststenotic dilation which had been interpreted as an aortic aneurysm in successive CT scans. The authors highlight the unusually late clinical presentation and misdiagnosis despite extensive radiologic investigation. The subarachnoid hemorrhage was probably a disease manifestation, since berry aneurysms are among the noncardiac malformations associated with AC.
A coartação da aorta (CA) representa ∼7% das cardiopatias congénitas sendo habitualmente diagnosticada na infância ou no início da vida adulta, dependendo da gravidade da obstrução e das malformações associadas. Não tratados, menos de 20% dos doentes sobrevivem até aos 50 anos. Descreve-se um caso de CA torácica, diagnosticada aos 61 anos, numa mulher com HTA conhecida desde os 45. Aos 56 anos foi internada por hemorragia subaracnoideia e, aquando da realização de angiografia cerebral, foi detetado aneurisma da aorta torácica. Quatro anos mais tarde foi referenciada à Consulta de Hipertensão por HTA não controlada e insuficiência cardíaca. O ecocardiograma mostrava hipertrofia ventricular esquerda e a HTA mantinha-se não controlada apesar de polimedicada. Aos 61 anos, por queixas de fadiga muscular intensa dos membros inferiores, efetuou Doppler Arterial que identificou índices de pressão sistólica diminuídos, bilateralmente e de forma simétrica, sugerindo estenose da aorta abdominal. A angio-RMN permitiu o diagnóstico de CA torácica com importante dilatação pós-estenótica, que foi interpretada em TAC seriadas como correspondendo a aneurisma da aorta descendente. Destaca-se a apresentação clínica tardia e diagnóstico erróneo, apesar de extensa investigação radiológica. Salienta-se ainda que a hemorragia subaracnoideia foi, provavelmente, uma manifestação da doença já que os aneurismas do polígono de Willis são uma das malformações extracardíacas associadas à CA.
Aortic coarctation is a relatively common abnormality that occurs in approximately 6–8% of patients with congenital heart disease.1 It can occur in isolation – simple coarctation – or in association with other cardiac and/or noncardiac lesions – complex coarctation.2 The most common intracardiac lesions (present in approximately 50% of cases) are patent ductus arteriosus (closely related to the proposed pathogenesis of AC3), ventricular and atrial septal defects, and bicuspid aortic valve. Chromosome abnormalities and prematurity are the most common noncardiac conditions associated with AC. Extracardiac vascular abnormalities are also common, including anomalies of the brachiocephalic artery and its branches – stenosis or aneurysm – and aneurysms of the circle of Willis (berry aneurysms).2
The clinical features depend largely on the nature of the associated cardiac lesions and severity of the narrowing. Diagnosis is usually made during childhood or early adult life; AC is almost always evident on physical examination and classic radiologic findings. However, left untreated, as the collateral circulation develops the typical clinical signs may disappear. The young adult with uncorrected coarctation is usually asymptomatic. The most frequent manifestation is hypertension (especially systolic hypertension) and, later on, congestive heart failure and coronary artery disease.
In an autopsy series of uncorrected coarctation, 50% of patients had died by age 30 and 90% by age 60.4 The most common causes of death are aortic rupture or dissection, cerebral hemorrhage due to aneurysm rupture and congestive heart failure. The former usually occurs before age 30, while congestive heart failure is a late consequence of long-standing hypertension and accelerated coronary artery disease.
Case reportIn 2001, a 56-year-old Caucasian woman, obese, with hypertension since age 45 and no other relevant medical history, was admitted to the emergency room of our hospital with a subarachnoid hemorrhage. Cerebral angiography was performed and incidentally disclosed the presence of a thoracic aortic aneurysm (7cm long, 6cm maximum diameter), below the aortic arch. After discharge the patient was referred to the vascular surgery outpatient clinic for follow-up.
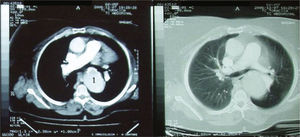
Over the course of three years, several computed tomography (CT) scans were performed, which showed no progression of the aneurysm (Figure 1).
The hypertension was controlled with a diuretic and the ECG was normal, with no evidence of left ventricular hypertrophy (LVH).
However, in 2005, the hypertension became uncontrolled and the patient developed symptoms of heart failure (exertional dyspnea, fatigue, orthopnea) and was referred to the outpatient hypertension clinic. On physical examination she was obese (BMI 35kg/m2), with high blood pressure (170/100mmHg) in the upper limbs and unmeasurable in the lower limbs due to extreme obesity, and tachycardic, with irregular arrhythmic pulse; cardiopulmonary auscultation revealed arrhythmic heartbeats (with no murmurs) and inspiratory bibasal crackles; and lower limb edema was evident. On initial complementary workup the ECG revealed atrial fibrillation and voltage criteria of LVH, and the echocardiogram disclosed LVH, non-dilated LV with preserved systolic function, mild aortic regurgitation (tricuspid aortic valve) and normal right-side chambers and valves; laboratory tests were normal, including renal function; the renal ultrasound was also normal. Despite treatment with several antihypertensive drug combinations, including ACE inhibitors, ARBs, chlorthalidone, furosemide, spironolactone, beta-blockers and calcium antagonists, her blood pressure remained uncontrolled. At that time another CT was performed which showed no progression of the aortic aneurysm.
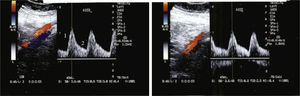
In 2007, her blood pressure remained uncontrolled and the patient complained of intense bilateral lower limb muscular fatigue, without calf pain. Arterial Doppler ultrasound was performed that revealed a symmetrically decreased ankle/brachial pressure index and persistent blood flow during diastole, suggesting abdominal aortic stenosis (Figure 2).
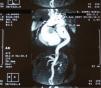
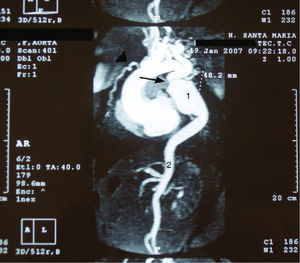
MRI angiography disclosed a thoracic aortic coarctation with large poststenotic dilation (7cm long, 4.8cm maximum diameter), hypoplastic abdominal aorta and collateral circulation via the left subclavian artery and internal mammary artery (Figure 3).
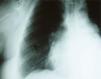
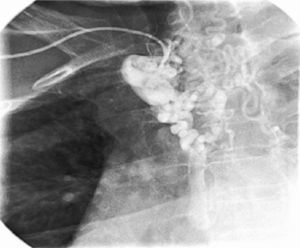
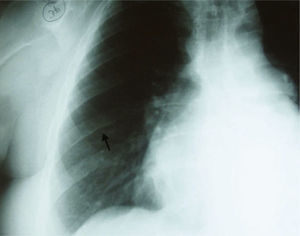
In order to exclude coronary artery disease (for surgical purposes) coronary angiography was also performed which revealed, once again, marked collateral circulation via the internal mammary artery and no coronary lesions (Figure 4). On retrospective review of the chest radiography, inferior rib notching was evident (Figure 5).
The patient was refused for surgery due to the high surgical risk imposed by the large and long poststenotic dilation.
Discussion and conclusionsThe striking feature of this case is, in our opinion, the very late clinical presentation (in pediatrics a diagnosis of AC is considered late when it occurs after the first year of age). This patient was probably born with mild AC, which passed unnoticed and allowed the progressive development of collateral circulation. Apart from high blood pressure, the subarachnoid hemorrhage was probably the first manifestation of the disease, as aneurysms of the circle of Willis (berry aneurysms) are among the most common noncardiac malformations associated with AC (five times more frequent than in the general population).5,6 The large poststenotic dilation is probably the consequence of a long-standing pressure gradient and intrinsic aortic medial tissue abnormalities (fragmentation of elastin, increase in ground substance and a reduction in smooth muscle cells) that are typically present in the coarctation segment and surrounding areas.7
The 2008 ACC/AHA guidelines for adults with congenital heart disease recommend intervention for AC in the following circumstances8:
- 1.
Peak-to-peak coarctation gradient greater than or equal to 20mmHg. The peak-to-peak gradient is a measurement derived from catheterization data in which the peak pressure beyond the AC is subtracted from the peak pressure proximal to the AC. However it is reasonable to use a maximum instantaneous gradient (by echocardiography) or measured BP differential in place of the peak-to-peak coarctation gradient.
- 2.
Peak-to-peak coarctation gradient less than 20mmHg in the presence of anatomic imaging evidence of significant coarctation with radiological evidence of significant collateral flow (which is responsible for the attenuation of the pressure gradient across the coarctation).
Despite her uncontrolled blood pressure, the patient is hemodynamically compensated due to the marked collateral circulation that has developed. In this situation of long-standing AC, regional blood flow is generally maintained within normal limits by autoregulatory vasoconstriction in the hypertensive areas and vasodilation in the hypotensive areas.9 In this case, hypertension is primarily mechanical due to flow obstruction. In addition, renal hypoperfusion may lead to enhanced renin secretion and subsequent volume expansion and arteriolar vasoconstriction. The only effective treatment is surgical repair or percutaneous angioplasty. However, even if the coarctation was repaired, it might not prevent persistence or late recurrence of systemic hypertension or disproportionate systolic hypertension during exercise.8,10 In a review, approximately 50% of patients undergoing repair after age 40 remained hypertensive.10 On the other hand, some reports show blood pressure improvements so that antihypertensive drugs are either discontinued or substantially reduced after the intervention in almost all patients.11
Conflicts of interestThe authors have no conflicts of interest to declare.