Contrast-induced nephropathy (CIN) is a life-threatening complication after primary percutaneous coronary intervention (p-PCI). Oxidative stress and inflammation may play an important role in the development of CIN.
ObjectiveWe aimed to assess the relationship between total oxidant status, total antioxidant capacity, high-sensitivity C-reactive protein (hs-CRP), gamma-glutamyltransferase and uric acid (UA) in the development of CIN in patients presenting with ST-elevation myocardial infarction (STEMI).
MethodsThis prospective cohort study consisted of 341 patients with STEMI. Patients were divided into two groups: those with and those without CIN. Predictors of CIN were determined by multivariate regression analyses.
ResultsMultivariate regression analysis showed that initial glucose level, contrast media volume/glomerular filtration ratio (eGFR) ratio, hs-CRP, UA and oxidative status index were associated with the development of CIN in patients with STEMI.
ConclusionThe main finding of this study is that increased oxidative stress and inflammation parameters were associated with the development of CIN in patients with STEMI. Other independent predictors of CIN were contrast media volume/eGFR ratio, initial glucose level, UA and hs-CRP.
A nefropatia induzida por contraste (NIC) é uma complicação crítica após intervenção coronária percutânea primária (ICP-p). O stress oxidativo e a inflamação podem ter um papel importante no desenvolvimento da NIC.
ObjetivoAvaliar a associação entre o estado oxidante total (EOT), a capacidade total (COT), a proteína C-reativa de alta sensibilidade (PCR-as), a gama glutamiltransferase (GGT) e o ácido úrico (AU) no desenvolvimento de doentes com NIC que se apresentam com STEMI.
MétodosEste estudo de coorte prospetivo incluiu 341 doentes com enfarte do miocárdio com elevação do segmento ST (STEMI). Os doentes foram divididos em dois grupos: os portadores e os não portadores de NIC. Os valores preditivos de NIC foram determinados por análises de regressão multivariada.
ResultadosA análise de regressão multivariada demonstrou que o nível inicial de glicose, o volume dos meios de contraste/taxa de filtração glomerular estimada (TFGe), a PCR-as e o índice do estado oxidativo foram associados ao desenvolvimento da NIC em doentes com STEMI.
ConclusãoO aumento do stress oxidativo e dos parâmetros inflamatórios estão associados ao desenvolvimento da NIC em doentes com STEMI. Outros fatores preditivos independentes da NIC foram o volume dos meios de contraste/TFG, o nível daglicemia inicial, o AU e a PCR-as.
Contrast-induced nephropathy (CIN) is a rare but life-threatening complication of primary percutaneous coronary interventions (pPCI). Preventing the development of CIN is very important for decreased mortality and morbidity after p-PCI. Our study demonstrated that oxidative parameters such as gamma-glutamyltransferase, 4 (UA), total oxidant status and total antioxidant status, as well as inflammatory parameters such as C-reactive protein could be useful prognostic biomarkers in acute myocardial infarction (AMI). Additionally, measurement of the oxidative status may be useful in preventing the development of CIN after pPCI.
IntroductionPrimary percutaneous coronary intervention (p-PCI) is the recommended reperfusion strategy for patients who present acute ST-elevation myocardial infarction (STEMI) within the first few hours after the onset of symptoms.1 However, some life-threatening complications such as contrast-induced nephropathy (CIN) are observed after p-PCI.2,3 Evidence shows a strong correlation between CIN and high mortality and morbidity in patients with STEMI.2 Additionally, these patients tend to be hospitalized for long periods.2 Several clinical and laboratory variables have been associated with the development of CIN, including contrast media volume, presence of diabetes mellitus, chronic congestive heart failure, anemia and decreased renal perfusion.3 Production of reactive oxygen species (ROS) and impaired renal perfusion are the two major mechanisms involved in the pathogenesis of CIN.4,5 In addition, previous studies have suggested that CIN may lead to a decrease in the activity of antioxidant enzymes and cause cytotoxic effects via the release of ROS. Increased reactive oxygen radicals may cause an increase in renal medullary hypoxia.4–6 Additionally, antioxidant supplements such as ascorbic acid and drugs such as N-acetylcysteine (NAC) could lead to the amelioration of CIN.7,8 Although many studies have shown alterations in total oxidant status and antioxidant capacity in various diseases, including end-stage renal disease, atrial fibrillation and hypertension, to the best of our knowledge, none have examined these parameters in predicting CIN in patients with STEMI.9–11
In a clinical setting, serum creatinine level is generally used as a marker of renal function, even though this marker is affected by several factors such as age, gender, muscle mass, weight, and ethnicity.12,13 Additionally, serum creatinine is not a marker of renal damage per se and is elevated only at advanced stages of renal disease. For these reasons, use of serum creatinine as a marker could lead to under-estimation of CIN.13 Patients at high risk of CIN should undergo early prophylactic measures such as hydration and the antioxidant NAC to prevent CIN.8 Additionally, high risk patients should be followed up for progression of serum creatinine levels after the procedure.2 Therefore, there is a need for the development of new laboratory parameters to predict the development of CIN. In this study, we aimed to assess the association between total oxidant status (TOS), total antioxidant capacity (TAC), high-sensitivity C-reactive protein (hs-CRP), gamma-glutamyltransferase (GGT) and uric acid (UA) in the development of CIN in patients presenting with STEMI.
Material and methodsStudy populationIn this prospective single-center study, 341 patients undergoing primary percutaneous coronary intervention (p-PCI) for diagnosis of STEMI were screened between June 2012 and July 2019. Exclusion criteria included hyperthyroidism (six patients), age <18-years, end-stage renal failure (15 patients), patients treated with emergent coronary artery bypass graft surgery (five patients), sepsis (five patients), exposure to contrast injection in the seven days prior to p-PCI (10 patients), known malignancy, severe hepatic dysfunction, and inflammatory disease. Patients who presented with cardiogenic shock or died during the first 72 hours of their hospital stay or during revascularization were also excluded from the study. The final study cohort consisted of 300 patients with STEMI. The study protocol was reviewed and approved by the local ethics committee in accordance with the Declaration of Helsinki. All participants provided written informed consent.
All p-PCIs were performed via the femoral artery by an experienced interventional cardiologist using the appropriate equipment (Toshiba Medical Systems, Nasushiobara, Japan). Non-ionic low osmolality contrast medium (Omnipaque 350 MG/ml; GE Healthcare, Cork, Ireland) was used for the p-PCIs and the total contrast medium volume used was recorded for all patients. All patients were administered 300 mg acetylsalicylic acid (ASA) with a loading dose of 300 mg clopidogrel before the procedure. A dose of 70–100 U/kg unfractionated heparin was administered after visualizing arterial anatomy. The use of bare metal or drug-eluting stents and glycoprotein IIb/IIIa was at the discretion of the physician. All of the patients received intravenous hydration with isotonic saline and were transferred to the intensive care unit (ICU) after the procedure, where treatment was continued with 100 mg ASA and 75 mg clopidogrel. The decision for concurrent use of statins, angiotensin-converting enzyme inhibitors and beta-blockers (BB) was made according to recommendations in the guidelines. After the coronary intervention, transthoracic echocardiography assessments were performed at the coronary ICU using (iE33, Philips Medical System, Andover, MA) with a 3.5 MHz transducer.
Diagnosis of contrast-induced nephropathyCIN is defined as an impairment of renal function and was measured as either a 25% increase in serum creatinine from baseline or a 0.5 mg/dL increase in absolute value when there was no alternative etiology within 72 hours after the first procedure.3
Diagnosis of ST-elevation myocardial infarctionDiagnoses were recorded by the participating physicians based on clinical, electrocardiographic and biochemical (elevated troponin levels) criteria. The type of myocardial infarction (ST-elevation vs. non-ST-elevation) and unstable angina were homogeneously defined and all patients were treated according to the currently available guidelines. p-PCI was performed in all patients.14
Risk scoring of contrast-induced nephropathySeveral clinical and laboratory variables are used to define the development of CIN. Recently, Mehran et al.,3 reported a simple risk score (Mehran risk score) for CIN after PCI. We calculated Mehran risk scores and compared them with oxidative stress parameters and other study variables to predict CIN in the present study.
Blood sampling and analysesBlood samples were taken after at least 12 hours of fasting. Venous blood samples were obtained upon patient admission, drawn from a large ante-cubital vein without interruption of venous flow, using a 19-gauge butterfly needle connected to a plastic syringe. The contents of the syringe was transferred immediately to polypropylene tubes. These tubes were then centrifuged at 4000 rpm for min at 10–18°C. Supernatant plasma samples were stored in plastic tubes at -80°C until analysis. Glomerular filtration ratio was calculated using the Cockcroft-Gault Equation.15
Total antioxidant status, total oxidant status, and oxidative stress index analysesTotal antioxidative capacity levels were determined spectrophotometrically (Rel Assay Diagnostics, Gaziantep, Turkey). The method is based on the bleaching of the characteristic color of a more stable (2,2′-azino-bis[3-ethylbenzothiazoline-6-sulfonic acid]) radical cation by antioxidants.16 The assay has excellent precision and a coefficient of variation less than 3%. The results were expressed as mmol Trolox (Rel Assay) equivalent/L. Total oxidative status levels were measured spectrophotometrically (Relassay, Diagnostics, Gaziantep, Turkey). In this method, oxidants present in the sample oxidized the ferrous ion-o-dianisidine complex to ferric ion. The oxidation reaction was enhanced by glycerol molecules abundantly present in the reaction medium. The ferric ion produced a colored complex with xylenol orange in an acidic medium. The color intensity, which could be measured spectrophotometrically, was related to the total amount of oxidant molecules present in the sample.17 The assay was calibrated with hydrogen peroxide and the results were expressed in terms of micromolar hydrogen peroxide equivalent per liter (μmol H2O2 equivalent/L). The coefficient of variation for individual plasma samples was less than 3%. The levels of TAC and TOS were assayed in an Olympus AU 2700 autoanalyzer (Japan). The oxidative stress index (OSI) is defined as the ratio of TOS to TAC levels, expressed as a percentage. For the calculation of OSI, TAC units were represented as mmol/L, and the OSI value calculated according to the following formula: OSI (arbitrary unit)=TOS (mmol H2O2 equiv./L)/TAC (mmol Trolox equiv./L). Serum hs-CRP level was measured by a chemiluminescent immunometric assay within 12–24 h of admission using available commercial kits according to the manufacturer's instructions (Immulite 2000, Siemens Medical Solutions Diagnostics, NJ, US). The intra- and inter-assay coefficients of variation for hs-CRP were <8.7%.
Statistical analysisSPSS version 16.0 software package was used for statistical analyses in this study. Categorical variables were expressed as frequency (%) and compared using the Chi-squared test. Kolmogorov-Smirnov test was used to test the distribution of numeric variables; those with normal distribution were expressed as mean ± standard deviation and were compared with Student's t-test. Data without normal distribution were expressed as median (inter-quartile range of 25%-75% percentiles) and were compared with the Mann-Whitney U test. In all statistical analyses, p value <0.05 was statistically significant. Univariate analysis and backward conditional binary logistic regression were performed to estimate the odds ratio (OR) and 95% confidence interval (CI) for the prediction of CIN. Receiver operating characteristics (ROC) curve analysis was used to analyze the prognostic value of TAC, TOS and OSI for CIN, following STEMI. C-Statistic (area under the curve) was presented as a unified estimate of sensitivity and specificity according to the cut-off value that was obtained by a ROC curve analysis. The optimal cut-off value was defined as the value yielding the maximal Youden index, or the best combined sensitivity and specificity.18 C-Statistic (area under the curve) was presented as a unified estimate of sensitivity and specificity.
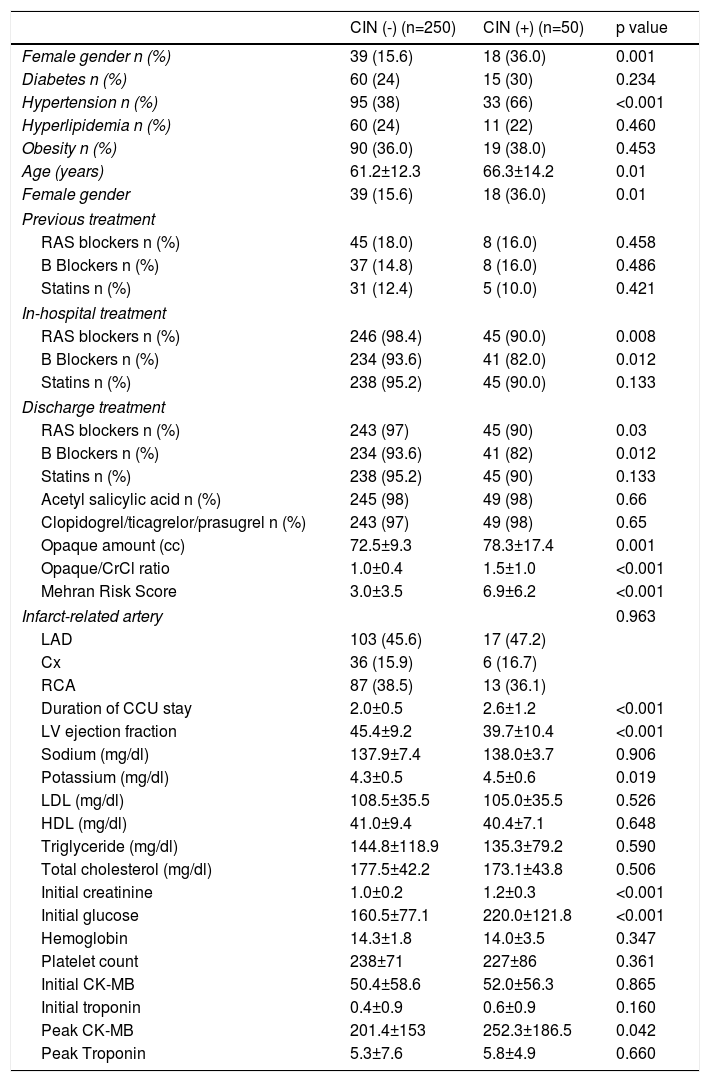
ResultsA total of 300 patients (mean age: 62±13 years; range, 23–91 years) were included in this study. During the follow-up period, 50 patients (16.7%) developed CIN. Demographic and clinical characteristics of the patients with and without CIN are listed in Table 1. The patients with CIN were significantly older, predominantly female and with a higher incidence of hypertension when compared to the patients without CIN (p=0.01, p=0.01 and p<0.001, respectively). The incidence of diabetes, obesity and hyperlipidemia were similar between patients with and without CIN (for all parameters p>0.05). There were no statistically significant differences in cholesterol parameters (p>0.05) between patients with and without CIN. Left ventricle ejection fraction (LVEF) was significantly lower in patients with CIN compared to patients without CIN (p<0.001). Initial creatinine levels were significantly higher in patients with CIN compared to patients without CIN.
Demographic and clinical characteristics of patients with and without contrast-induced nephropathy.
| CIN (-) (n=250) | CIN (+) (n=50) | p value | |
|---|---|---|---|
| Female gender n (%) | 39 (15.6) | 18 (36.0) | 0.001 |
| Diabetes n (%) | 60 (24) | 15 (30) | 0.234 |
| Hypertension n (%) | 95 (38) | 33 (66) | <0.001 |
| Hyperlipidemia n (%) | 60 (24) | 11 (22) | 0.460 |
| Obesity n (%) | 90 (36.0) | 19 (38.0) | 0.453 |
| Age (years) | 61.2±12.3 | 66.3±14.2 | 0.01 |
| Female gender | 39 (15.6) | 18 (36.0) | 0.01 |
| Previous treatment | |||
| RAS blockers n (%) | 45 (18.0) | 8 (16.0) | 0.458 |
| B Blockers n (%) | 37 (14.8) | 8 (16.0) | 0.486 |
| Statins n (%) | 31 (12.4) | 5 (10.0) | 0.421 |
| In-hospital treatment | |||
| RAS blockers n (%) | 246 (98.4) | 45 (90.0) | 0.008 |
| B Blockers n (%) | 234 (93.6) | 41 (82.0) | 0.012 |
| Statins n (%) | 238 (95.2) | 45 (90.0) | 0.133 |
| Discharge treatment | |||
| RAS blockers n (%) | 243 (97) | 45 (90) | 0.03 |
| B Blockers n (%) | 234 (93.6) | 41 (82) | 0.012 |
| Statins n (%) | 238 (95.2) | 45 (90) | 0.133 |
| Acetyl salicylic acid n (%) | 245 (98) | 49 (98) | 0.66 |
| Clopidogrel/ticagrelor/prasugrel n (%) | 243 (97) | 49 (98) | 0.65 |
| Opaque amount (cc) | 72.5±9.3 | 78.3±17.4 | 0.001 |
| Opaque/CrCl ratio | 1.0±0.4 | 1.5±1.0 | <0.001 |
| Mehran Risk Score | 3.0±3.5 | 6.9±6.2 | <0.001 |
| Infarct-related artery | 0.963 | ||
| LAD | 103 (45.6) | 17 (47.2) | |
| Cx | 36 (15.9) | 6 (16.7) | |
| RCA | 87 (38.5) | 13 (36.1) | |
| Duration of CCU stay | 2.0±0.5 | 2.6±1.2 | <0.001 |
| LV ejection fraction | 45.4±9.2 | 39.7±10.4 | <0.001 |
| Sodium (mg/dl) | 137.9±7.4 | 138.0±3.7 | 0.906 |
| Potassium (mg/dl) | 4.3±0.5 | 4.5±0.6 | 0.019 |
| LDL (mg/dl) | 108.5±35.5 | 105.0±35.5 | 0.526 |
| HDL (mg/dl) | 41.0±9.4 | 40.4±7.1 | 0.648 |
| Triglyceride (mg/dl) | 144.8±118.9 | 135.3±79.2 | 0.590 |
| Total cholesterol (mg/dl) | 177.5±42.2 | 173.1±43.8 | 0.506 |
| Initial creatinine | 1.0±0.2 | 1.2±0.3 | <0.001 |
| Initial glucose | 160.5±77.1 | 220.0±121.8 | <0.001 |
| Hemoglobin | 14.3±1.8 | 14.0±3.5 | 0.347 |
| Platelet count | 238±71 | 227±86 | 0.361 |
| Initial CK-MB | 50.4±58.6 | 52.0±56.3 | 0.865 |
| Initial troponin | 0.4±0.9 | 0.6±0.9 | 0.160 |
| Peak CK-MB | 201.4±153 | 252.3±186.5 | 0.042 |
| Peak Troponin | 5.3±7.6 | 5.8±4.9 | 0.660 |
Data presented as mean ± SD or number (%) of the patients.
CCU: coronary care unit; CIN: contrast-induced nephropathy; CK-MB: creatine kinase myocardial bundle; CrCl: creatinine clearance; Cx: circumflex; HDL: high density lipoprotein; LAD: left anterior descending artery; LDL: low density lipoprotein; LV: left ventricle; RAS: renin angiotensin system; RCA: right common artery.
There were no statistically significant differences in previous treatments including renin-angiotensin system (RAS) blockers, acetyl salicylic acid, clopidogrel or statins between patients with and without CIN (for all parameters p>0.05). Among in-hospital treatments, the use of BB and RAS blockers was lower in patients with CIN but use of other medications was similar among patients with and without CIN. Patients with CIN spent longer in the coronary care unit than patients without CIN (2.6±1.2 vs. 2.0±0.5 days; p<0.001). There were no statistically significant differences in the initial and peak cardiac enzymes including troponin and creatine kinase myocardial bundle between patients with and without CIN (for all parameters p>0.05). The mean Mehran risk score (6.9±6.2 vs. 3.0±3.5, p<0.001), contrast media volume (78.3±17.4 cc vs. 72.5±9.3 cc, p<0.001) and contrast media volume/glomerular filtration ratio (eGFR) (1.5±1.0 versus 1.0±0.4; p<0.001) were significantly higher in patients with CIN compared to patients without CIN.
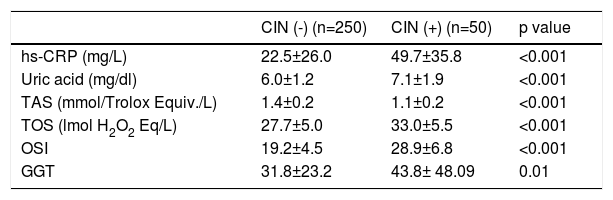
Oxidative status and inflammation parameters are presented in Table 2. In patients with CIN, plasma TOS and OSI values were significantly higher (33.0±5.5 mmol H2O2 equiv./L vs. 27.7±5.0 mmol H2O2 equiv./L, p<0.001 and 28.9±6.8 vs. 19.2±4.5, p<0.001, respectively) and plasma TAC levels were significantly lower compared to those without CIN (1.1±0.2 mmol Trolox equiv./L vs. 1.4±0.2 mmol Trolox equiv./L p<0.001). Hs-CRP, UA and GGT levels were significantly higher in patients with CIN compared to patients without CIN (49.7±35.8 mg/L vs. 22.5±26.0 mg/L, p<0.001; 7.1±1.9 mg/dl vs. 6.0±1.2 mg/dl, p<0.001 and 43.8±48.09 U/L vs. 31.8±23.2 U/L, p=0.01; respectively)
Comparison of plasma oxidative stress and inflammation parameters in patients with and without contrast-induced nephropathy.
| CIN (-) (n=250) | CIN (+) (n=50) | p value | |
|---|---|---|---|
| hs-CRP (mg/L) | 22.5±26.0 | 49.7±35.8 | <0.001 |
| Uric acid (mg/dl) | 6.0±1.2 | 7.1±1.9 | <0.001 |
| TAS (mmol/Trolox Equiv./L) | 1.4±0.2 | 1.1±0.2 | <0.001 |
| TOS (lmol H2O2 Eq/L) | 27.7±5.0 | 33.0±5.5 | <0.001 |
| OSI | 19.2±4.5 | 28.9±6.8 | <0.001 |
| GGT | 31.8±23.2 | 43.8± 48.09 | 0.01 |
Data presented as mean ± SD.
OSI: oxidative stress index; Hs-CRP: high-sensitivity C-reactive protein; TAS: total antioxidative status; TOS: total oxidative status.
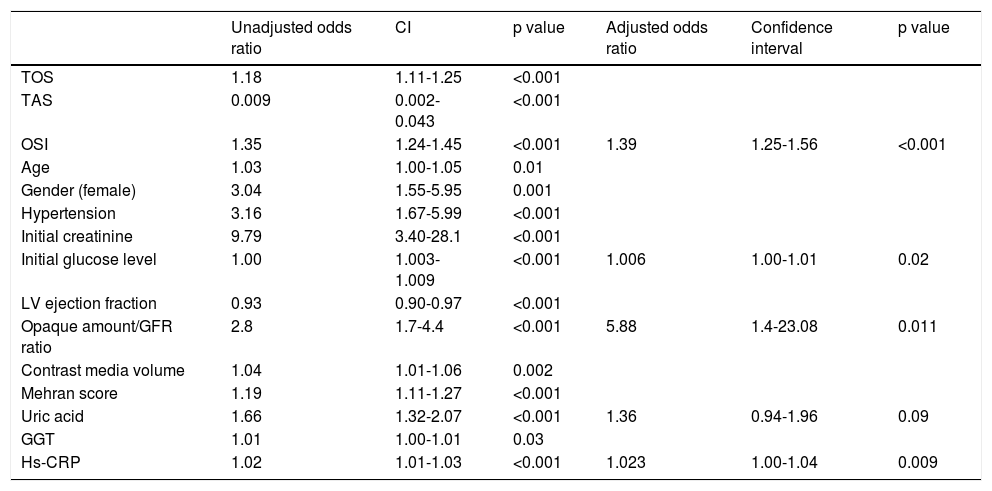
Univariate analyses showed that TOS, TAC, OSI, LVEF, age, contrast media volume, contrast media volume/eGFR ratio, female gender, hypertension, initial glucose level, initial creatine level, Mehran score, UA level, hs-CRP level and GGT level were significantly associated with a higher risk of development of CIN (Table 3).
Univariate and multivariate regression analyses of predictors of contrast-induced nephropathy in the study population.
| Unadjusted odds ratio | CI | p value | Adjusted odds ratio | Confidence interval | p value | |
|---|---|---|---|---|---|---|
| TOS | 1.18 | 1.11-1.25 | <0.001 | |||
| TAS | 0.009 | 0.002-0.043 | <0.001 | |||
| OSI | 1.35 | 1.24-1.45 | <0.001 | 1.39 | 1.25-1.56 | <0.001 |
| Age | 1.03 | 1.00-1.05 | 0.01 | |||
| Gender (female) | 3.04 | 1.55-5.95 | 0.001 | |||
| Hypertension | 3.16 | 1.67-5.99 | <0.001 | |||
| Initial creatinine | 9.79 | 3.40-28.1 | <0.001 | |||
| Initial glucose level | 1.00 | 1.003-1.009 | <0.001 | 1.006 | 1.00-1.01 | 0.02 |
| LV ejection fraction | 0.93 | 0.90-0.97 | <0.001 | |||
| Opaque amount/GFR ratio | 2.8 | 1.7-4.4 | <0.001 | 5.88 | 1.4-23.08 | 0.011 |
| Contrast media volume | 1.04 | 1.01-1.06 | 0.002 | |||
| Mehran score | 1.19 | 1.11-1.27 | <0.001 | |||
| Uric acid | 1.66 | 1.32-2.07 | <0.001 | 1.36 | 0.94-1.96 | 0.09 |
| GGT | 1.01 | 1.00-1.01 | 0.03 | |||
| Hs-CRP | 1.02 | 1.01-1.03 | <0.001 | 1.023 | 1.00-1.04 | 0.009 |
CI: confidence interval; GFR: glomerular filtration rate; GGT: gamma-glutamyltransferase; Hs-CRP: high-sensitivity C-reactive protein; LV: left ventricle; OSI: oxidative stress index; TAS: total anti-oxidative status; TOS: total oxidative status.
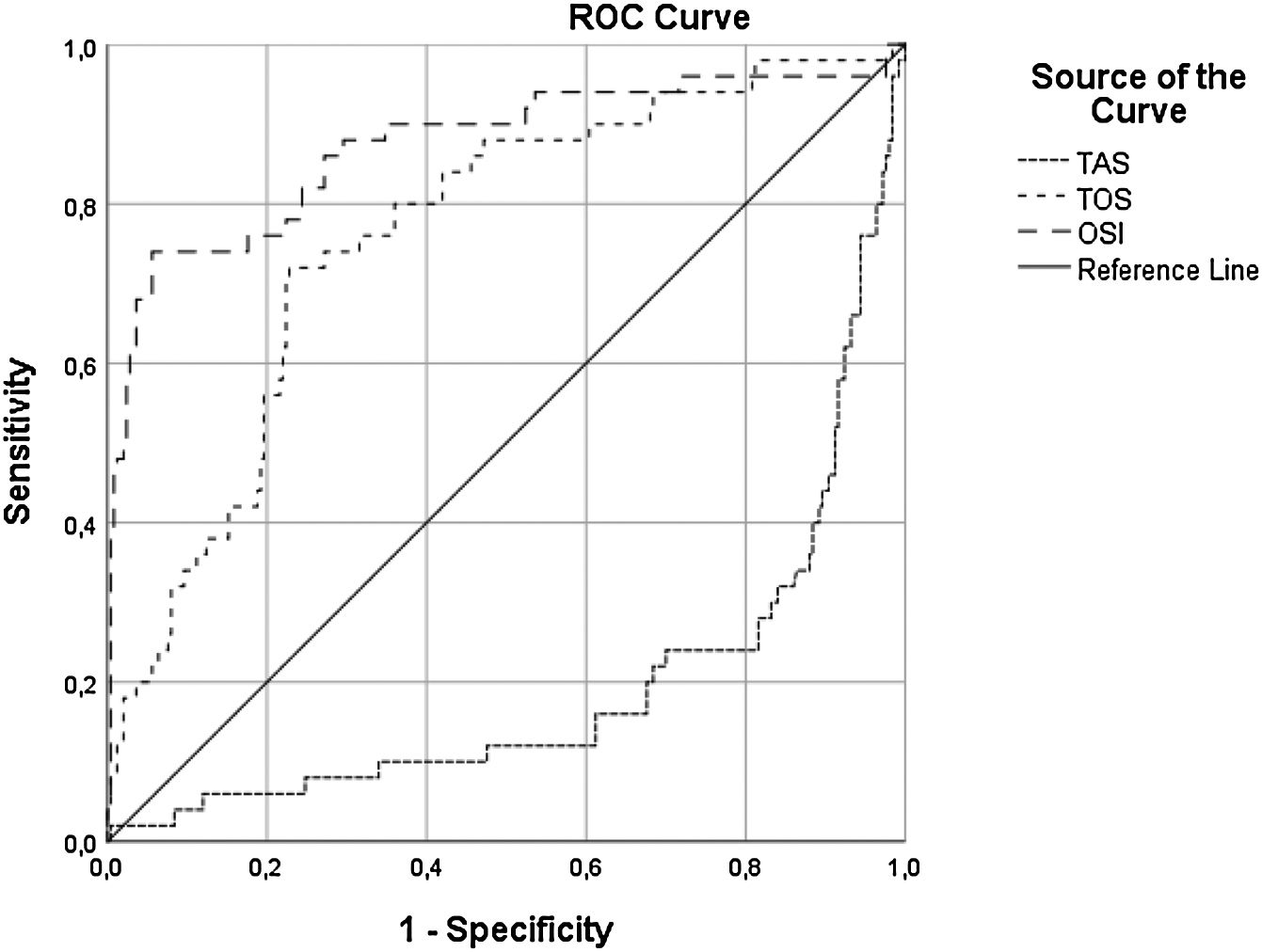
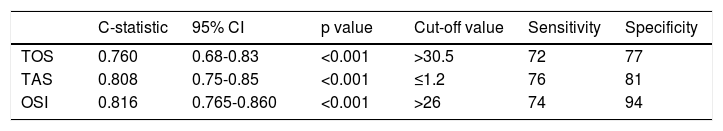
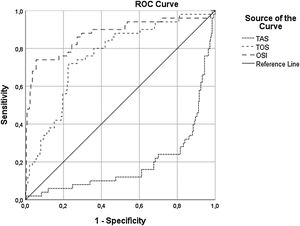
A multivariate binary logistic regression analysis was carried out by including all the parameters that were associated with the development of CIN in the univariate analysis. This analysis showed that OSI (OR: 1.39; CI 95: 1.25-1.56, p<0.001), initial glucose level (OR: 1.006; CI 95%: 1.00-1.01, p=0.02), hs-CRP (OR: 1.023; CI 95: 1.00-1.04, p=0.009), contrast media volume/eGFR ratio (OR: 5:88; CI 95%: 1.4-23.08, p=0.011), and UA level (OR: 1.36; CI 95%: 0.94-1.96, p=0.09) remained as independent factors for CIN development (Table 3). ROC curve analysis showed that OSI (C-statistic: 0.81; CI 95%: 0.76-0.86, p<0.001), TAC (C-statistic: 0.80; CI 95%: 0.75-0.85, p<0.001) and TOS (C-statistic: 0.76; CI 95%: 0.68-0.83, p<0.001) were significant predictors of CIN following STEMI (Table 4, Figure 1). We calculated that a cut-off point of 30.5 for TOS, 1.2 for TAC and 26 for OSI could estimate the presence of CIN with a sensitivity of 72%, 76% and 74%, a specificity of 77%, 81% and 94%, respectively (Table 4).
Receiver operating characteristics curve analysis of oxidative parameters.
| C-statistic | 95% CI | p value | Cut-off value | Sensitivity | Specificity | |
|---|---|---|---|---|---|---|
| TOS | 0.760 | 0.68-0.83 | <0.001 | >30.5 | 72 | 77 |
| TAS | 0.808 | 0.75-0.85 | <0.001 | ≤1.2 | 76 | 81 |
| OSI | 0.816 | 0.765-0.860 | <0.001 | >26 | 74 | 94 |
CI: confidence interval; OSI: oxidative status index; TAS: total anti-oxidative status; TOS: total oxidative status.
Receiver operating characteristics curve with calculated area under the curve and optimal cut-off point for oxidative stress index, total antioxidant capacity, and total oxidant status to identify the presence of contrast-induced nephropathy. OSI: oxidative stress index; TAS: total antioxidant status; TOS: total oxidant status.
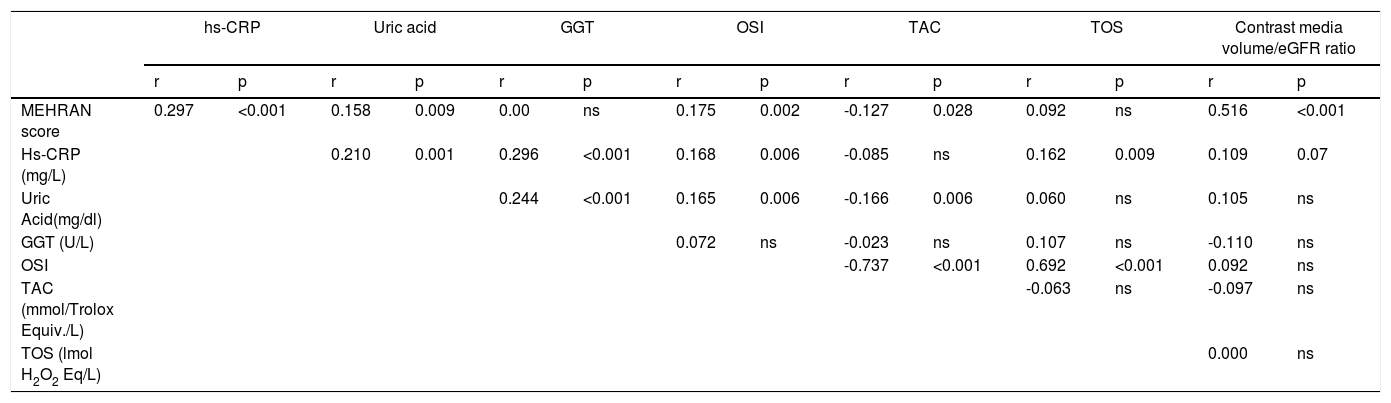
In correlation analysis, Mehran score correlated positively with OSI, hs-CRP, UA, contrast media volume/eGFR ratio levels and negatively correlated with TAC levels, while hs-CRP levels correlated positively with UA, GGT, OSI and TOS (Table 5).
Correlation analysis of study variables.
| hs-CRP | Uric acid | GGT | OSI | TAC | TOS | Contrast media volume/eGFR ratio | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| r | p | r | p | r | p | r | p | r | p | r | p | r | p | |
| MEHRAN score | 0.297 | <0.001 | 0.158 | 0.009 | 0.00 | ns | 0.175 | 0.002 | -0.127 | 0.028 | 0.092 | ns | 0.516 | <0.001 |
| Hs-CRP (mg/L) | 0.210 | 0.001 | 0.296 | <0.001 | 0.168 | 0.006 | -0.085 | ns | 0.162 | 0.009 | 0.109 | 0.07 | ||
| Uric Acid(mg/dl) | 0.244 | <0.001 | 0.165 | 0.006 | -0.166 | 0.006 | 0.060 | ns | 0.105 | ns | ||||
| GGT (U/L) | 0.072 | ns | -0.023 | ns | 0.107 | ns | -0.110 | ns | ||||||
| OSI | -0.737 | <0.001 | 0.692 | <0.001 | 0.092 | ns | ||||||||
| TAC (mmol/Trolox Equiv./L) | -0.063 | ns | -0.097 | ns | ||||||||||
| TOS (lmol H2O2 Eq/L) | 0.000 | ns | ||||||||||||
GGT: Gamma-glutamyltransferase; OSI: oxidative stress index; Hs-CRP: high-sensitivity C-reactive protein; ns: not statistically significant; TAC: total antioxidative capacity; TOS: total oxidative status.
The main findings of this study suggest that oxidative stress and inflammation parameters are associated with the development of CIN in patients presenting with acute STEMI. Oxidative stress is caused by increased ROS production and is associated with poor outcomes in cardiovascular diseases such as coronary artery disease (CAD) and CIN.4,9,19–22
Borekci et al.,20 demonstrated that OSI, UA and neutrophil to lymphocyte ratio were associated with spontaneous reperfusion in patients with STEMI. Similarly, Turan et al.21 reported that plasma TOS and OSI were significant factors associated with CAD complexity and severity in patients with acute coronary syndrome. Korkmaz et al.22 showed that thiol/disulfide homeostasis, a new oxidative marker, could be a good biochemical risk marker for CIN in STEMI patients who underwent p-PCI. Additionally, we have reported a positive association between atrial fibrillation development after STEMI and TAC, TOS and OSI.9 In these studies, the relationship between CIN and oxidative stress markers in patients with STEMI was not determined. Data from the current study suggest that plasma TAC, TOS and OSI were increased in patients with CIN when compared to those without CIN in a STEMI patient population. Thus, increased oxidative stress may contribute to pathogenesis in these patients.
CIN is a serious complication of p-PCI after STEMI and is associated with poor clinical outcomes such as prolonged hospital stays, rising costs, and increased short- and long-term morbidity and mortality.2,23 It is, therefore, important to estimate which patients may develop CIN. There are several established parameters for predicting CIN development. Yildiz et al.24 showed that serum osmolality is a predictive factor for CIN development, while Gohbara et al.25 determined that acidosis was associated with CIN development in patients with STEMI. Our results showed that OSI, initial glucose levels, contrast media volume/eGFR ratio, UA and hs-CRP levels were associated with CIN development in patients with STEMI after p-PCI. Previous studies have also shown similar results.26–28 Contrast volume and basal renal insufficiency are important risk factors for CIN29; accordingly, the contrast media volume/eGFR ratio is a good indicator for CIN.28 Our study also indicated that contrast media volume/eGFR ratio was a powerful predictor of CIN development.
There is a demonstrated association between inflammation and CIN in the literature.28,30–33 Liu et al.32 showed that an increase in hs-CRP was a significant and independent predictor of CIN in patients with STEMI after p-PCI. Oweis et al.34 showed that interleukin-33 levels were elevated in patients with CIN. Gu et al.33 reported that an elevation of the inflammatory biomarkers hs-CRP and procalcitonin was a risk factor for postoperative CIN. Similarly, in the present study, hs-CRP levels were significantly higher in patients with CIN compared to patients without CIN. This indicates that higher hs-CRP may be associated with the development of CIN in patients presenting with STEMI.
Although several studies have shown that pharmacological agents such as theophylline, fenoldopam, mannitol, iloprost, furosemide, dopamine, hemofiltration, ascorbic acid, NAC and sodium bicarbonate (NaHCO3) could prevent the development of CIN, it is still unclear which are the best agents for the prevention of CIN.35 Among these agents, the use of NAC and NaHCO3 with physiological saline was recommended as a good strategy to prevent CIN.35 Additionally, statins that have pleiotropic effects such as a decrease in free oxygen radicals, increase in nitrous oxide production and increment of vascular smooth muscle relaxation, can also prevent CIN.36 The present study supported the beneficial effects of the above mentioned agents. In future studies, there is a need to focus on agents and foods with antioxidant effects; individuals should also be advised to increase the amount of antioxidants in their diets.
In this study we have demonstrated lower antioxidant capacity and higher oxidative and inflammatory status in STEMI patients. During the acute phase of STEMI, the oxidative and inflammatory response may influence the patient's baseline cardio-renal reserves. The addition of oxidative and inflammatory parameters to the current risk models derived from clinical, angiographic, and laboratory-based variables and to the previously validated Mehran risk score13 may result in significant improvement in the prediction of CIN.
Our study has several limitations: the study population consisted of patients treated with different modes of STEMI and the number of CIN patients was too small for definitive conclusions. Additional oxidative stress parameters such as serum prolidase activity, malondialdehyde, superoxide dismutase etc., were not examined, and markers for the detection of renal failure, such as NGAL, cystatin C, urinary Kim-1 were also not analyzed.
ConclusionsThe present study suggests that in a patient population with STEMI, oxidative stress and inflammation were significantly higher in patients with CIN when compared to patients without CIN. Further studies are needed to establish the pathophysiological and clinical significance of increased oxidative stress and inflammation, and to investigate the effect of antioxidant and anti-inflammatory agents in patients with AMI.
Conflicts of interestThe authors have no conflicts of interest to declare.