Cisplatin induces many collateral effects such as gastrointestinal disorders, nephrotoxicity, and dysautonomia. Recently our group showed that cisplatin treatment induces gastric emptying delay and that physical exercise and treatment with pyridostigmine prevent this change. In the current study, we investigated the role of moderate exercise on cardiac activity and autonomic balance in rats treated with cisplatin.
MethodsMale Wistar rats were divided into saline, cisplatin, exercise, and exercise+cisplatin groups. Cardiac and autonomic disorders were induced by (cisplatin – 3 mg/kg, i.p. once a week/per 5 weeks). Exercise consists of swimming (1 hour per day/5× day per week/per 5 weeks without overload). Forty-eight hours after the last session of the training or treatment, we assessed the cardiac activity and HRV via electrocardiogram analysis in DII derivation.
ResultsCisplatin increase (p<0.05) R–R′ interval and decrease (p<0.05) heart rate vs. saline. Exercise+cisplatin prevented (p<0.05) changes in R–R′ interval. Exercise per se induced bradycardia vs. saline group. We observed an increase in LF (nu) and a decrease in HF (nu) in the cisplatin group vs. saline. These changes were not significant. Moreover, cisplatin treatment increased (p<0.05) QT, QTc, and JT intervals compared with the saline group. In the exercise+cisplatin groups these increases were prevented significantly (p<0.05).
ConclusionIn the current study, chronic use of cisplatin induced electrocardiographic changes without altering autonomic balance. Moderate physical exercise prevented this phenomenon indicating that exercise can be beneficial in patients in chemotherapy.
O uso crónico de cisplatina induz diversos efeitos colaterais, como distúrbios gastrointestinais, nefrotoxicidade e disautonomia. Recentemente o nosso grupo demonstrou que o tratamento com cisplatina induz diminuição do esvaziamento gástrico e que o exercício físico bem como o tratamento com piridostigmina previnem essa alteração. No presente estudo, investigamos o papel do exercício moderado sobre a cardiotoxicidade e no equilíbrio autonómico em ratos tratados com cisplatina.
MétodosUtilizamos ratos machos wistar divididos em: Controlo, Cisplatina, Exercício e Exercício + Cisplatina. A cardiotoxicidade foi induzida por (Cisplatina 3mg/kg, i.p 1x/semana/5 semanas). O exercício consiste em natação (1-h/dia/5x/semana/5 semanas sem sobrecarga); 48 horas após a última sessão do treinamento e/ou tratamento, avaliamos a atividade cardíaca e a VFC através da análise de ECG na derivação DII.
ResultadosCisplatina aumenta (P < 0,05) intervalo R-Ŕ e diminui (P < 0,05) FC versus ratos controlo. Exercício + Cisplatina previne (P < 0,05) as alterações no intervalo R-Ŕ. Exercício per si induz bradicardia versus grupo controlo. Observamos aumento de LF (nu) e diminuição de HF (nu) no grupo cisplatina versus controlo. No entanto, essas mudanças não foram significativas. Além disso, o tratamento com cisplatina aumentou (P < 0,05) os intervalos QT, QTc e JT em comparação com o grupo controlo. Os grupos exercício + cisplatina previnem significativamente (P < 0,05) esses aumentos.
ConclusãoNo presente estudo, o uso crónico de cisplatina induz alterações no electrocardiograma sem modificar o balanço autonómico nos grupos estudados. O exercício físico de intensidade moderada preveniu esse fenómeno, indicando que o exercício pode ser benéfico em pacientes em quimioterapia.
Cisplatin is the most widely prescribed drug for chemotherapy and is effective in treating diverse human cancers.1 However, cisplatin has several clinical side effects, including nausea, emesis, anorexia, weight loss, disrupted gastrointestinal function, diarrhea, and delayed gastric motility.2–4
Cisplatin is primarily associated with nephrotoxicity and its dose-limiting side effects. Nephrotoxicity causes acute kidney injury and chronic kidney disease associated with intracellular stresses, including DNA damage, mitochondrial pathology, oxidative stress, and endoplasmic reticulum stress, as well as ototoxicity.5,6 Moreover, acute and cumulative cisplatin treatment induces cardiotoxicity, a limiting factor of chemotherapy.7
Various studies have indicated that cisplatin induces an array of cardiotoxic events, occurring during or shortly after infusion, such as symptomatic arrhythmias, angina, myocarditis, pericarditis, diastolic disturbances, cardiac ischemia, and acute myocardial infarction.8 Consequently, various pathogenic pathways have been suggested to allay cisplatin-induced cardiotoxicity, such as oxidative damage and programmed cell death/apoptosis.
Autonomic dysfunction is defined as an imbalance of the autonomic nervous system, leading to dysfunction and changes in the cardiovascular system, such as cardiotoxicity, hypertension, and decreased baroreflex sensitivity.9–11 Moreover, clinical studies have indicated that cisplatin can induce autonomic dysfunction in patients, where the most common adverse effects are fatigue, nausea, vomiting, postural hypotension12,13; and cardiovascular damage.14,15
To mitigate the adverse effects caused by cisplatin, physical exercise can be a non-pharmacological intervention to reduce the adverse effects of chemotherapy.16 In this regard, physical exercise seems to act on the autonomic nervous system, stimulate the vagus nerve via the parasympathetic nervous system, reduce sympathetic excitability, and maintain the sympathovagal balance.17 Furthermore, chronic use of cisplatin has been associated with the development of muscle atrophy by increasing levels of muscle RING finger-1 (MuRF1) and atrogin-1. Physical exercise has, therefore, been proven to be capable of preventing muscle atrophy by inducing muscle protein synthesis and hypertrophy, enhancing the positive effects of the Akt/mTOR/p70S6 kinase pathway.18 However, little is known about the effects of moderate physical exercise on cisplatin-induced cardiotoxicity.
ObjectivesThe study aimed to assess whether moderate physical exercise can modulate autonomic balance and improve changes in ECG parameters induced by chronic use of cisplatin in rats.
MethodsAnimals and ethical approvalMale Wistar rats weighing between 250+30 g from the Federal University of Piau, Brazil were used. The animals were housed in collective cages with water and feed ad libitum, temperature control (25±2°C), and 12/12 h light/dark cycle. All procedures were performed according to the recommendations of the “Guide to the Care and Use of Laboratory Animals and approved by the Ethics Committee for Animal Use (CEUA) of Federal University of Piauí, Brazil (Protocol 431/18)”. Four groups were formed: saline, cisplatin, exercise, and exercise+cisplatin.
Cisplatin treatmentCisplatin (Citoplax® 50 mg/50 ml, Pharmaceutical Chemical Laboratory Bergamo Ltda S/A, São Paulo, Brazil) was administered at a dose of 3 mg/kg via i.p. once per week, for 5 weeks, according to Silva et al.19. The control rats received only 0.9% saline solution via s.c. However, all groups treated with cisplatin received 2 ml of the 0.9% saline to prevent the nephrotoxicity induced by chemotherapy.20
Moderate physical exercise protocolThe exercise protocol was adapted from Lima et al.21 Initially, all rats underwent a period of adaptation to the liquid medium before training. Physical exercise was started simultaneously with the cisplatin or control treatment. The exercise consisted of swimming with an overload of 5% body weight attached to the tail of the rats for a period of 1 hour per day, 5 days a week for 5 weeks. The sedentary rats were subjected to contact with shallow water, not requiring exercise effort, to eliminate any stress bias caused by water contact.
Cardiac activity (ECG) and HRV assessmentTwenty-four hours after the last cisplatin treatment and/or the last physical exercise session, rats were anesthetized with ketamine (80 mg/kg i.m.) and xylazine (20 mg/kg, i.m.) for the bilateral implant of electrodes in the thoracic region and right hind paw for electrocardiogram (ECG) monitoring via DII derivation. Twenty-four hours after surgery, the rats were connected to an ECG module connected to a Bridge Amp computerized biological signal acquisition system (Powerlab 4/20, ADInstruments, LabChart Pro 8.0) for continuous ECG monitoring, and measurement of the PR interval and recording of the corrected QT, QTc, JT, QRS and PR values. Their heart rate (HR, bpm) was recorded during ECG through the R–R′ interval in addition to the autonomic study through evaluation of heart rate variability (HRV). To determine the QTc interval, Bazett's formula was used, as follows:
Statistical analysisInitially, the data were analyzed for normal distribution by the Shapiro–Wilk test. For comparisons between three or more groups, we used one-way analysis of variance (ANOVA) followed by the Tukey post hoc test. The results are expressed as mean±SEM. Values of p<0.05 were considered statistically significant.
ResultsIn this study, we observed that cisplatin treatment induced changes in electrocardiogram and bradycardia in rats. Exercise prevented these changes in ECG. Table 1 reports the results of the autonomic system by the components LF, HF, LF/HF, and RMSSD of HRV. We did not observe changes between the groups.
Representative values of the HRV (low frequency – LF (normative unit (nu and %)), high frequency – HF (normative unit (nu and %)), LF/HF ratio, and RMSSD) in rats control, cisplatin, exercise, and exercise+cisplatin. Data are expressed as the mean±SEM of 7 animals per group.
| LF (nu) | LF (%) | HF (nu) | HF (%) | LF/HF | RMSSD | |
|---|---|---|---|---|---|---|
| Saline | 23.4±0.9 | 19.6±0.7 | 61.0±0.9 | 49.3±2.7 | 0.36±0.02 | 17.8±4.3 |
| Cisplarin | 33.5±10.2 | 16.7±1.2 | 47.6±7.9 | 41.6±10.9 | 0.45±0.16 | 15.7±6.2 |
| Exercise | 22.0±4.8 | 13.0±2.0 | 64.3±3.8 | 47.5±8.5 | 0.38±0.10 | 9.2±2.9 |
| Exercise+cisplarin | 25.4±1.8 | 17.3±1.4 | 62.0±2.4 | 45.4±5.2 | 0.38±0.05 | 20.4±5.5 |
nu: normative unite; RMSSD: root mean square of the successive differences.
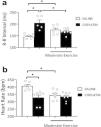
As depicted in Figure 1a, the cisplatin treatment significantly increased (p<0.05) the R–R interval compared with the saline group, while moderate exercise significantly prevented (p<0.05) these increases in the R interval. As shown in Figure 1b, cisplatin treatment significantly induced bradycardia (p<0.05) in comparison with the saline group. However, moderate exercise plus cisplatin did not affect this alteration in ECG compared to the cisplatin group.
Effect of physical exercise for 5 weeks on the R–R′ interval, and heart rate (bpm) in saline, cisplatin, exercise, or exercise+cisplatin rats. Differences were evaluated using one-way ANOVA with Tukey post hoc comparisons; values of *p<0.05 were considered significant (n=5–8 rats for group).
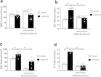
According to Figure 2a, no difference was observed between the saline and cisplatin groups in the QRS interval. On the other hand, Figure 2b–d shows that cisplatin induced an increase (p<0.05) in the QT, QTc, and JT intervals compared to the saline group. In all situations, moderate exercise plus cisplatin prevented these increases (p<0.05) in comparison with the cisplatin group.
Effect of physical exercise for 5 weeks on the QRS interval, QT interval, QTc interval, and JT interval in rats saline, cisplatin, exercise, and exercise+cisplatin. Differences were evaluated using one-way ANOVA with Tukey post hoc comparisons; values of *p<0.05 were considered significant (n=5–8 rats for group).
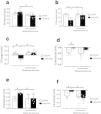
According to Figure 3a, no difference was observed between the saline and cisplatin groups in the PR interval. But in the group with moderate exercise plus cisplatin, we observed a decrease (p<0.05) in the PR interval compared to the cisplatin group. Regarding the P wave duration, the cisplatin treatment induced a significant decrease (p<0.05) compared with the saline group. Moderate exercise plus cisplatin significantly prevented (p<0.05) this decrease compared with the cisplatin group (Figure 3b).
Effect of physical exercise for 5 weeks on the PR interval, P duration, T, Q, R, and S amplitude, in rats saline, cisplatin, exercise, and exercise+cisplatin. Differences were evaluated using one-way ANOVA with Tukey post hoc comparisons; values of *p<0.05 were considered significant (n=5–8 rats for group).
Figure 3c illustrates the reduction (p<0.05) in ST height with cisplatin treatment in comparison with the saline group. Moreover, we observed that moderate exercise plus cisplatin significantly prevented (p<0.05) this decrease compared with the cisplatin group. Figure 3d and f indicates that cisplatin treatment induced a decrease (p<0.05) in the Q and S amplitude compared with the saline group. Moderate exercise plus cisplatin significantly prevented this decrease in the Q and S amplitude wave (p<0.05) compared with the cisplatin group. According to Figure 3e, cisplatin increased (p<0.05) the R wave amplitude compared with the saline group. No differences between moderate exercise plus cisplatin and cisplatin only were observed.
In Figure 4, we present an original representation trace of the four groups (control, cisplatin, exercise, and exercise+cisplatin) and a 3D image of all groups. There was a significant difference (p<0.05) between the QT interval waves in the cisplatin groups compared with the control group. In this sense, we observed that exercise+cisplatin prevented (p<0.05) these changes in electrocardiography induced by cisplatin.
DiscussionIn this study, the chronic use of cisplatin was found to promote cardiotoxicity and various changes in the electrocardiogram parameters. We first noted that this phenomenon was prevented by moderate exercise, an important non-pharmacological tool.
Platinum-induced peripheral neurotoxicity is a common side effect of platinum-based chemotherapy, which can prompt patients to reduce the dose or discontinue treatment, with oxaliplatin being more neurotoxic.22 Cisplatin or cis-diamminedichloroplatinum (II) (Cl2H6N2Pt+2) is a synthetic inorganic and water-soluble platinum complex in which the two ammine and two chloride ligands are oriented in a cis planar configuration around the central platinum ion.23,24 Cisplatin-induced toxicity is rare, and its pathophysiology is unknown. However, in a case report, the authors observed high-degree atrioventricular (AV) block, confirmed by ECG.25
The current study showed that cisplatin caused many changes in cardiac functions, such as modifications in the ECG pattern when compared to normal ECG. These changes were in agreement with Hu et al.,7 and were represented by changes in HR, RR, and QRS intervals. ECG of cisplatin rats also showed prolonged QT, QTc, and JT duration and a decrease in ST segment compared to normal ECG, which implies abnormal ventricular repolarization.26
Cisplatin induces dysautonomia, increases sympathetic tonus and decreases parasympathetic vagal tonus.19 In this study, cisplatin treatment produced an increase in blood pressure and oxygen consumption, reduction in coronary perfusion time, decreased oxygen supply by the myocardium, and caused tissue damage. This tissue injury can lead to cardiac remodeling, which may have contributed to the decrease in HR after the use of cisplatin.
We observed that cisplatin-induced bradycardia associated with increases in R–R′ interval. Moderate exercise did not influence changes in HR, but prevented an increase in the R–R′ interval. This HR and R–R′ interval behavior has been widely described in the literature, associated with physical exercise adaptations to the cardiovascular system. However, we only observed improvements in the R–R′ interval in cisplatin rats who did exercise, without changes in HR. In a previous study by our group, we found that cisplatin decreased intrinsic HR and that exercise prevented this phenomenon. We suggest that this decrease occurs due to high cardiac effort induced by sympathetic hyperactivity, which indicates tissue damage and cardiac remodeling.27 In rats with cisplatin exercise, there was no change. Furthermore, we did not observe changes in HRV components (LF, HF, LF/HF, and RMSSD) in rats treated with cisplatin.
We suggest that the response to the dose and time of administration can be influenced by these parameters. Cardiotoxicity is not frequently observed due to cisplatin treatment. However, some authors have reported a large array of cardiotoxic events occurring after cisplatin infusion.7 These include silent and symptomatic arrhythmias, angina, myocarditis, pericarditis, diastolic disturbances, cardiac ischemia, and acute myocardial infarction.
The mechanisms of cisplatin's cardiotoxic activity are still unknown. Some hypotheses regarding cisplatin-induced bradycardia have been suggested, such as interference in normal myocardial conduction, which can potentially influence the QT interval.28 Moreover, cisplatin is suspected of interfering with the regulation of action potential in myocyte sodium channels, like congenital forms of long QT syndrome. Cardiac arrhythmias resulting from QT prolongation can be severe and life-threatening. A longer QT interval was associated with a higher risk of Torsades de Pointes, potentially leading to sudden cardiac death.29
Regarding the arrhythmic spectrum, cisplatin has also been reported to induce bradycardia. In the clinical cases where bradycardia has been reported during or shortly after cisplatin infusion, with no disturbances detected in the patient's serum electrolyte concentrations, blood pressure or other physical parameters.30 Electrolyte disturbances are common causes of QT prolongation and include hypokalemia, hypomagnesemia, hypocalcemia, and even hypoglycemia.31 In this study, we observed that chronic treatment with cisplatin induced changes in electrocardiogram parameters with QT, QTc and JT interval prolongation, which were suggestive of cardiotoxicity. Moreover, this study showed that moderate exercise can be used associated with chemotherapy to prevent cisplatin-induced cardiotoxicity.
Several studies have described the effects of regular physical exercise in preventing or mitigating changes that occur in the autonomic mechanisms of cardiovascular control.32 Since autonomic dysfunction is one of the adverse effects of cisplatin administration, we suggest that the increase in blood pressure may be associated with the balance of autonomic function.19 In addition, there is evidence of better baroreflex sensitivity in physically active individuals compared to sedentary ones.
Cisplatin treatment can stimulate cell autophagy in fast-twitch skeletal muscles by downregulating the Akt and phosphorylated FOXO3a signaling pathways. On the other hand, both ladder and aerobic exercise directly affect muscle wasting by modulating the AKT/PGC1-α/FOXO3a signaling pathways, regardless of the skeletal muscle type, and can prevent muscle atrophy by altering the autophagy signaling pathway.33 Moreover, the protective effect of exercise in the cisplatin-treated rats was associated with reduced endothelial cell lesions and increased renal production of NO in trained rats.34 In this sense, high-intensity interval protocols promoted renoprotective actions, because they were more effective in mitigating cisplatin-induced acute kidney injury, in part by modulation of important markers of the innate immune response.34
ConclusionIn summary, our results demonstrate that moderate physical exercise contributes to attenuate cisplatin-induced cardiac disorders, as shown by the electrocardiogram results. However, further studies are needed to clarify the underlying molecular and cellular mechanisms involved in these effects.
LimitationsThis study used only an electrocardiogram as the cardiac evaluation parameter. However, other more objective parameters, such as echocardiography and cardiac morphology, could have been performed to confirm possible functional changes and morphological changes in structures after the use of chemotherapy drugs and physical exercise. Another point of this study is that analysis of rats can bring highly variable results. Furthermore, the data were acquired at different voltages. Future studies should, therefore, seek to minimize these limitations.
Conflicts of interestThe authors have no conflicts of interest to declare.
This study was part of an MSc Dissertation on Pharmacology presented by Mariana Sousa Silva to the Graduate Program in Pharmacology, Federal University of Piauí, Teresina, PI, Brazil.