Chagas disease is an endemic disease in Latin America that is increasingly found in non-endemic areas all over the world due to the flow of migrants from Central and South America. We present the case of a Brazilian immigrant in Portugal who underwent orthotopic heart transplantation for end-stage Chagas cardiomyopathy. Immunosuppressive therapy included prednisone, mycophenolate mofetil and tacrolimus. Twelve months after the procedure she is asymptomatic, with good graft function, and with no evidence of complications such as graft rejection, opportunistic infections, neoplasms or reactivation of Trypanosoma cruzi infection. By reporting the first case in Portugal of heart transplantation for Chagas cardiomyopathy, we aim to increase awareness of Chagas disease as an emerging global problem and of Chagas cardiomyopathy as a serious complication for which heart transplantation is a valuable therapeutic option.
A doença de Chagas é uma doença endêmica da América Latina cada vez mais reconhecida em áreas não endêmicas por todo o mundo, devido ao crescente fluxo migratório proveniente da América Central e do Sul. Apresentamos o caso de uma mulher de nacionalidade brasileira imigrante em Portugal submetida a transplante cardíaco por miocardiopatia de Chagas. A terapêutica imunossupressora a que foi submetida incluiu prednisona, micofenolato mofetil e tacrolimus. Doze meses após o procedimento a doente está assintomática, apresenta boa função do enxerto e sem evidência de complicações no seguimento, como rejeição, infeções oportunistas, neoplasias ou reativação da infeção por Trypanosoma cruzi. Ao reportar o primeiro caso em Portugal de transplante cardíaco por miocardiopatia de Chagas pretendemos alertar para o reconhecimento da doença de Chagas como um problema emergente em nível global e da miocardiopatia de Chagas como uma grave complicação da doença, para a qual o transplante cardíaco é uma válida opção terapêutica.
Chagas disease is emerging as a health care concern in non-endemic countries due to increasing emigration of Latin Americans. Chagas cardiomyopathy (CCM) is a severe complication of the disease and an established indication for heart transplantation (HTx). Because this condition is frequently neglected outside Latin America as a cause of chronic heart failure (CHF), awareness needs to be raised among health care professionals in non-endemic countries.
The present article reports the first HTx for CCM in Portugal and its outcome during the first year of follow-up, and reviews current practice of graft management in Chagas heart transplant recipients.
Case reportA 56-year-old female Brazilian immigrant in Portugal with end-stage CCM underwent HTx. She had been treated with nifurtimox at some point and had an implanted cardioverter-defibrillator (ICD) for secondary prevention. In the 12 months before HTx, she had multiple hospital admissions for CHF decompensation and had episodes of syncope due to ventricular tachycardia, without delivery of shocks by the ICD. In January 2015, she was admitted to a heart failure care unit in New York Heart Association (NYHA) functional class IV, with left ventricular ejection fraction (LVEF) of 19%, peak oxygen consumption of 12 ml/kg/min and cardiac index of 1.15 l/min/m2. She also had hyponatremia (131 mmol/l) and elevated brain natriuretic peptide (1112 pg/ml). She was under maximum tolerated doses of beta-blockers, angiotensin-converting enzyme inhibitors, mineralocorticoid receptor antagonists and digoxin. At this stage, the local heart team decided to include her on the HTx list and she received a graft from a 35-year-old male donor on February 7, 2015.
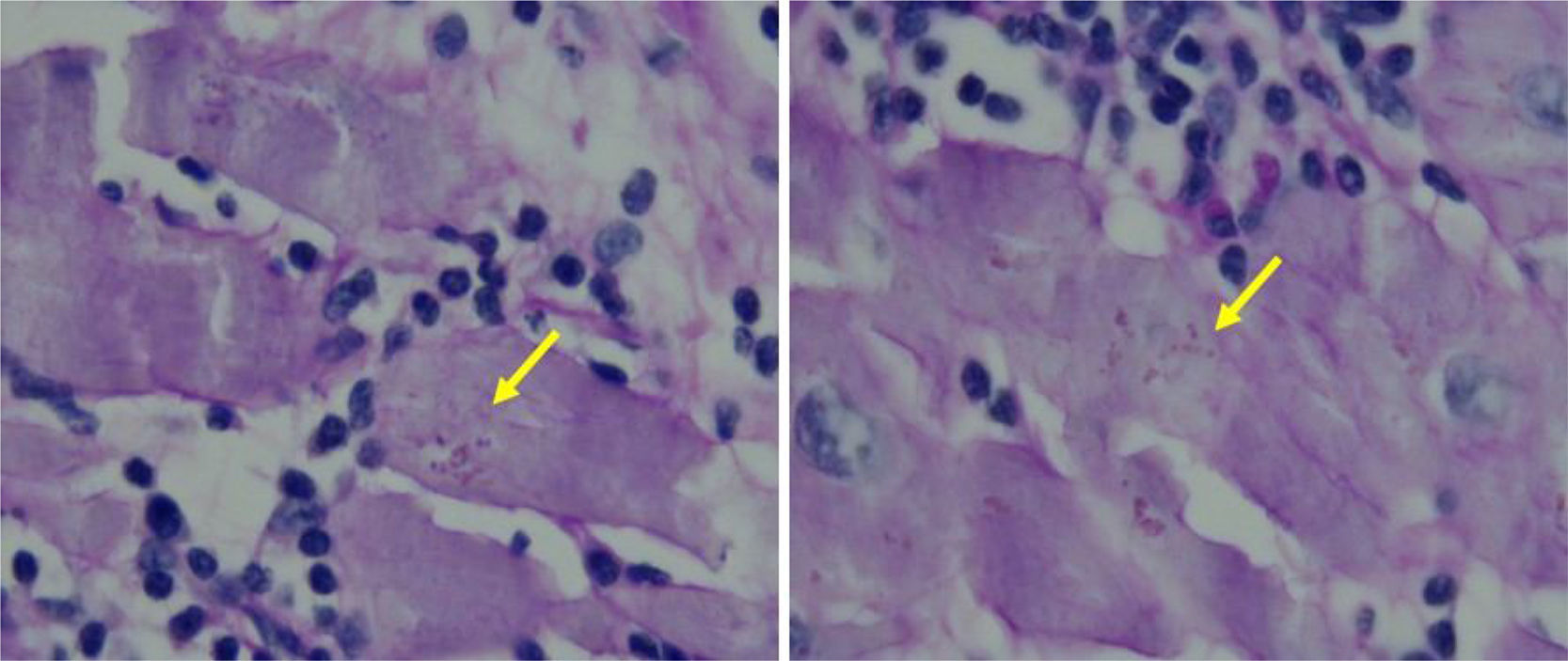
Postoperative recovery was uneventful. The immunosuppressive scheme included prednisone, tacrolimus and mycophenolate mofetil (MMF) in the usual dosage. There was no evidence of early rejection or Trypanosoma cruzi infection reactivation and she was discharged 12 days after surgery. Microscopic examination of the explanted heart revealed intracytoplasmic amastigotes (Figure 1), patchy fibrosis and an apical aneurysm.
Twelve months after HTx the patient is in NYHA class I and has resumed a normal life, with good graft function. Monitoring for T. cruzi reactivation included clinical assessment for signs or symptoms of cardiac, neurological or cutaneous involvement, electrocardiography to detect arrhythmias or conduction disturbances, echocardiography to detect left ventricular dysfunction, whole blood testing by polymerase chain reaction (PCR) for T. cruzi reactivation (performed every two months), and endomyocardial biopsy (EMB) to screen for amastigotes, as suggested.1 No episodes of T. cruzi reactivation occurred or were suspected. Prophylactic antiparasitic therapy was not used. She has had 10 EMBs, as per protocol, which showed no evidence of graft rejection. There have been no opportunistic infections or evidence of neoplasm.
DiscussionChagas disease, caused by the protozoan hemoflagellate T. cruzi, develops in three stages: acute, usually asymptomatic (although fever and chills may occur); indeterminate, with persistent parasitemia but no organ involvement; and chronic (after 10-30 years), characterized by cardiac and/or digestive tract involvement.2
Cardiomyopathy results from slowly progressive chronic myocarditis, associated with focal hypoperfusion due to abnormalities of coronary microvasculature, leading to dilatation of all four chambers, formation of typical apical aneurysms and damage to conductive tissue. CHF associated with CCM carries higher mortality than other more frequent causes of cardiac failure3 and is the most frequent cause of chronic systolic dysfunction in endemic countries.4 Most patients (two-thirds) die from sudden cardiac death, refractory CHF (25-30%) or thromboembolism (10-15%).5
Antiparasitic therapy (with nifurtimox or benznidazole) may be curative in the acute stage of the disease6; during the indeterminate stages it may prevent progression to chronic aggressive forms, as well as congenital transmission.7 In the chronic stage, benznidazole effectively reduces parasite detection but has no impact on clinical progression in patients with established CCM.8
HTx is the only approach to symptomatic CCM that appears to have benefit, with better outcomes compared to other more frequent indications for the procedure.9 NYHA class IV, LVEF <30%, inotropic dependence, digoxin use or no beta-blocker use, hyponatremia, peak oxygen consumption <10 ml/kg/min and need for ≥4 appropriate ICD shocks within 30 days are all predictors of one-year survival of less than 30%, and have been proposed as selection criteria for HTx.10 In the present case, NYHA class, LVEF, hyponatremia and digoxin therapy contributed to our patient's poor prognosis and were taken into account in the decision to perform HTx as the best therapeutic approach, which we believe may have been the first in Portugal.
Survival in the first month after HTx for CCM has been reported to be as high as 83%, without complicating rejection or infection in 53% and 57% of cases, respectively.11 Parasitemia may occur in up to 29% of patients, while T. cruzi reactivation has rarely been observed.12 None of these situations occurred in our patient in early follow-up.
There is no consensus as to the ideal immunosuppressive regimen for Chagas HTx recipients, and conventional schemes using triple combinations have been used according to local protocols, as in our case. Overall, there are no major differences in rejection rate between Chagas and non-Chagas HTx recipients,10 and one can speculate that conventional schemes of immunosuppressors for this special cohort of patients may be quite acceptable. Opportunistic infections seem to occur less often in Chagas transplant recipients than in other HTx recipients,13 while the incidence of malignancies in HTx recipients is similar.13,14 In our case, serial EMBs showed no evidence of rejection, and no opportunistic infections or signs of malignancy were detected during the first year of follow-up.
Recurrence of Chagas disease is a major concern after HTx because of the risk of allograft dysfunction, and was once considered a contraindication for HTx. Since the work of Bocchi et al., published in 1996,11 the success and benefit of HTx for CCM patients have become clear, and today it is a frequent indication for HTx worldwide, the leading indication in some endemic countries. Immunosuppressive therapy increases the risk of T. cruzi reactivation, particularly steroids used in high-dose pulses for graft rejection.14–16 Furthermore, MMF appears to lead to higher frequency and earlier occurrence of reactivation episodes compared to azathioprine-based protocols.10,12,13,17 Cyclosporine seems to play no role in reactivation of T. cruzi infection.10 Although it has been suggested that the combination of the more potent immunosuppressive agents, tacrolimus and MMF, is associated with higher rate of T. cruzi reactivation,9 our experience using this strategy in this patient has been good so far.
Nevertheless, T. cruzi reactivation is easily treated using benznidazole (5 mg/kg/day for two months) or allopurinol (600-900 mg/day for two months), and the overall prognosis is good (reported mortality of 0.7%9). Screening for reactivation should be based on PCR of whole blood samples, which is sufficiently sensitive to detect reactivation before complications can develop, enabling timely treatment.
Finally, medium- and long-term survival after HTx in Chagas patients is surprisingly good (with survival rates at one, two, six and 10 years as high as 83%, 76%, 62% and 46%, respectively10) and some series indicate a greater benefit of the procedure compared to non-Chagas HTx recipients.9
ConclusionCCM is emerging as a cause of CHF and arrhythmic complications outside Latin America. Health care workers should be aware of this disease and have a high degree of suspicion when dealing with Latin American immigrants presenting with a cardiomyopathy of unknown cause. For those with end-stage HF of Chagas etiology, antiparasitic drugs can no longer affect their poor outcome. HTx should be regarded as a viable and beneficial therapeutic option for these patients, with a greater survival benefit compared to non-Chagas HTx recipients. The feared T. cruzi infection reactivation in graft recipients is no longer a contraindication for HTx, is easily treated with antiparasitic drugs, and portends a very low mortality rate.
Conflicts of interestThe authors have no conflicts of interest to declare.