Zolmitriptan is a drug used in the acute treatment of migraine, which should not be used in high cardiovascular risk individuals because of its potential to induce vasospasm.
We report a rare case of myocardial infarction precipitated by taking zolmitriptan.
O zolmitriptano é um fármaco usado no tratamento agudo da enxaqueca, que pelo potencial de induzir vasoespasmo não deve ser utilizado em indivíduos com risco cardiovascular elevado, doença cardíaca isquémica (DCI) e ou história de vasoespasmo coronário.
Relatamos o caso raro de um enfarte agudo do miocárdio (EAM) precipitado pela toma de zolmitriptano.
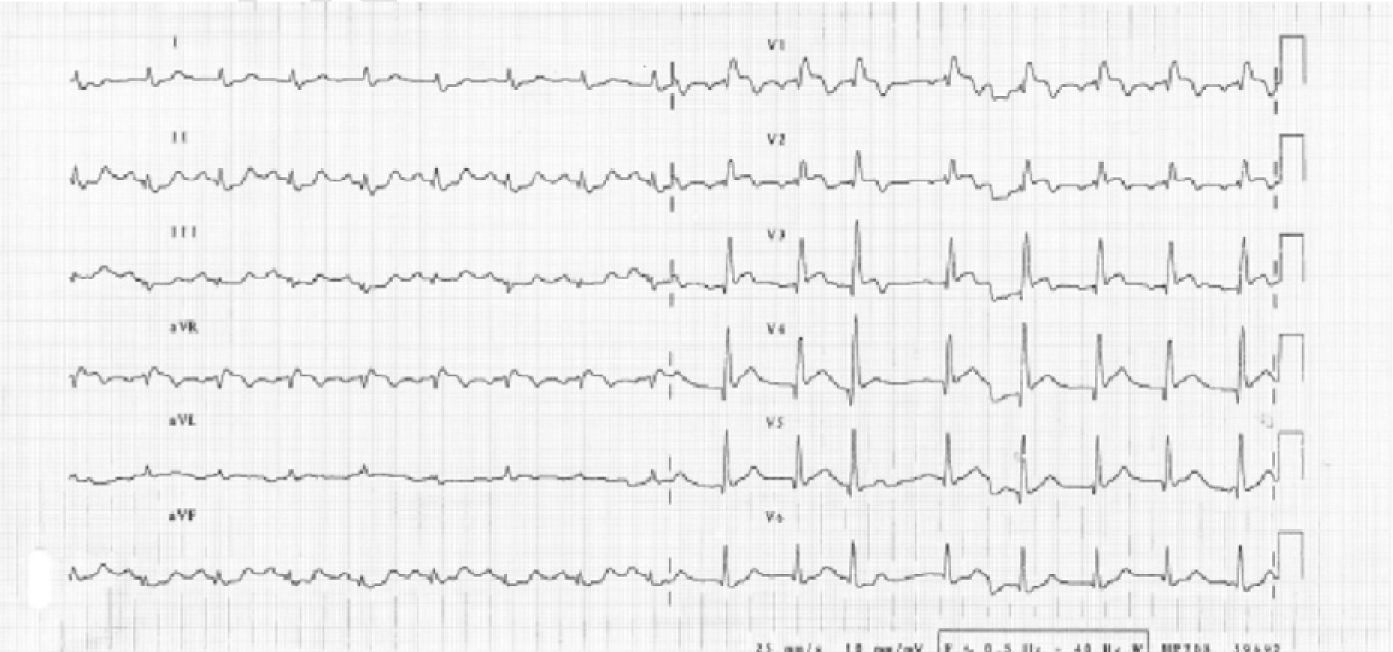
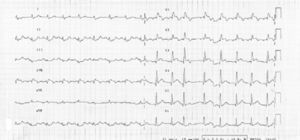
We present the case of a 73-year-old man with the following vascular risk factors: untreated hypertension and dyslipidemia and a history of stroke 14 years previously with minor sequelae. He presented at the Emergency Department (ED) with headache and was prescribed aspirin and zolmitriptan and discharged home. Thirty minutes after taking the drugs, he experienced persistent constricting chest pain and, still symptomatic, returned to the ED, where the electrocardiogram showed sinus rhythm, Q waves suggestive of ST-segment elevation in V1–V6 and ST-segment depression in DII, DIII and aVF (Fig. 1).
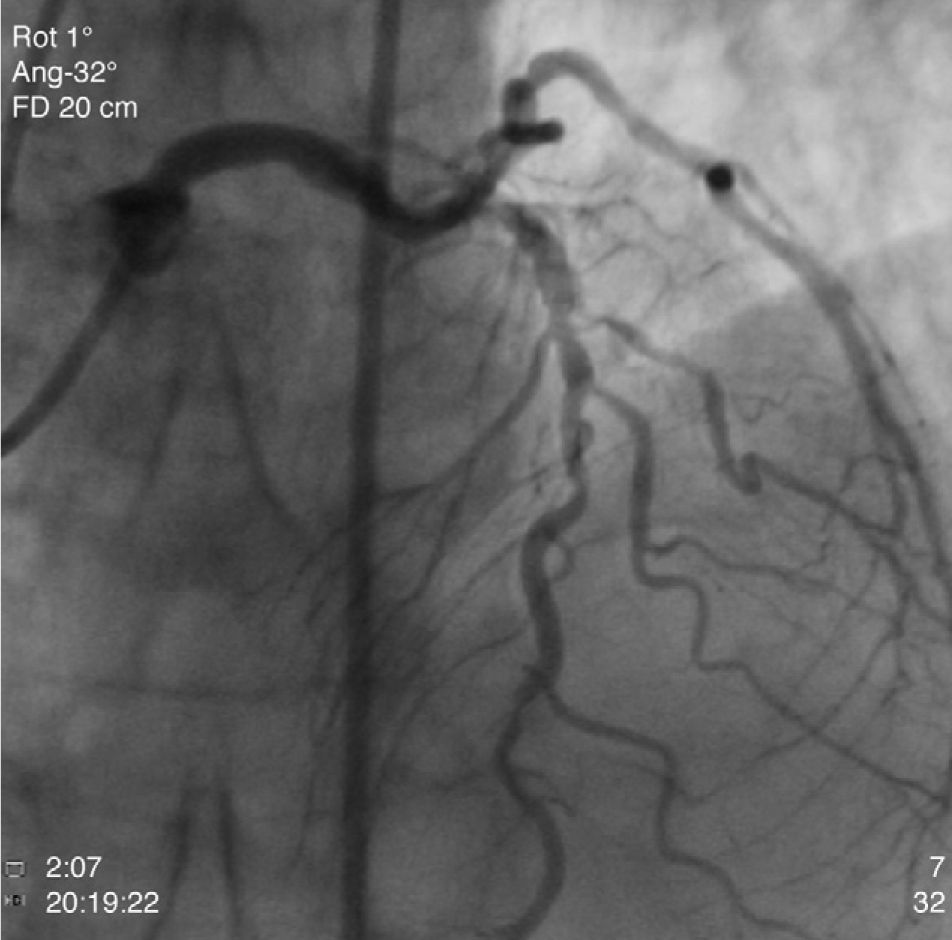
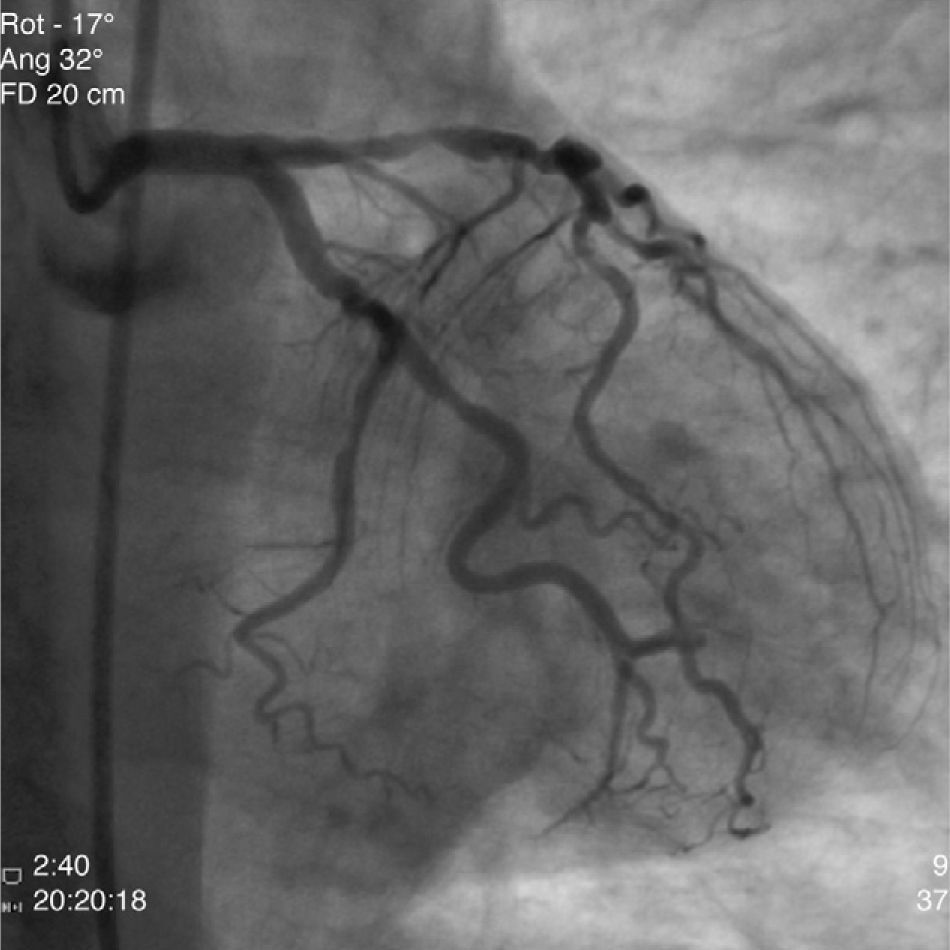
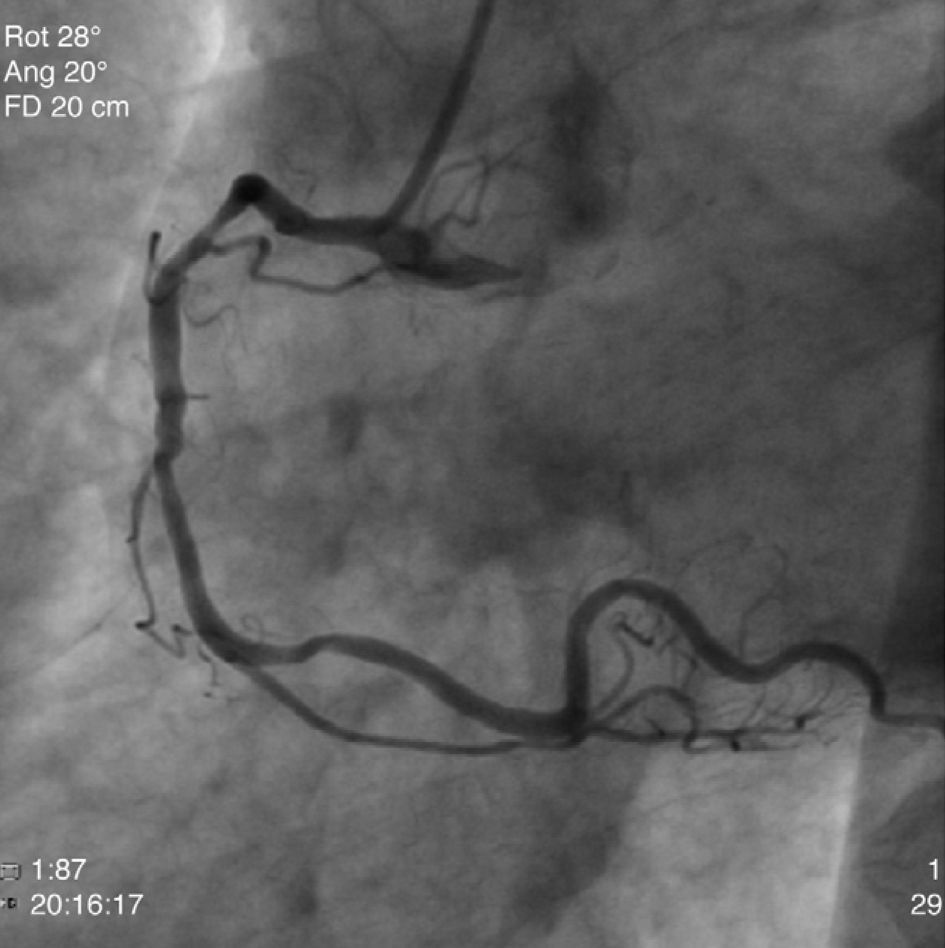
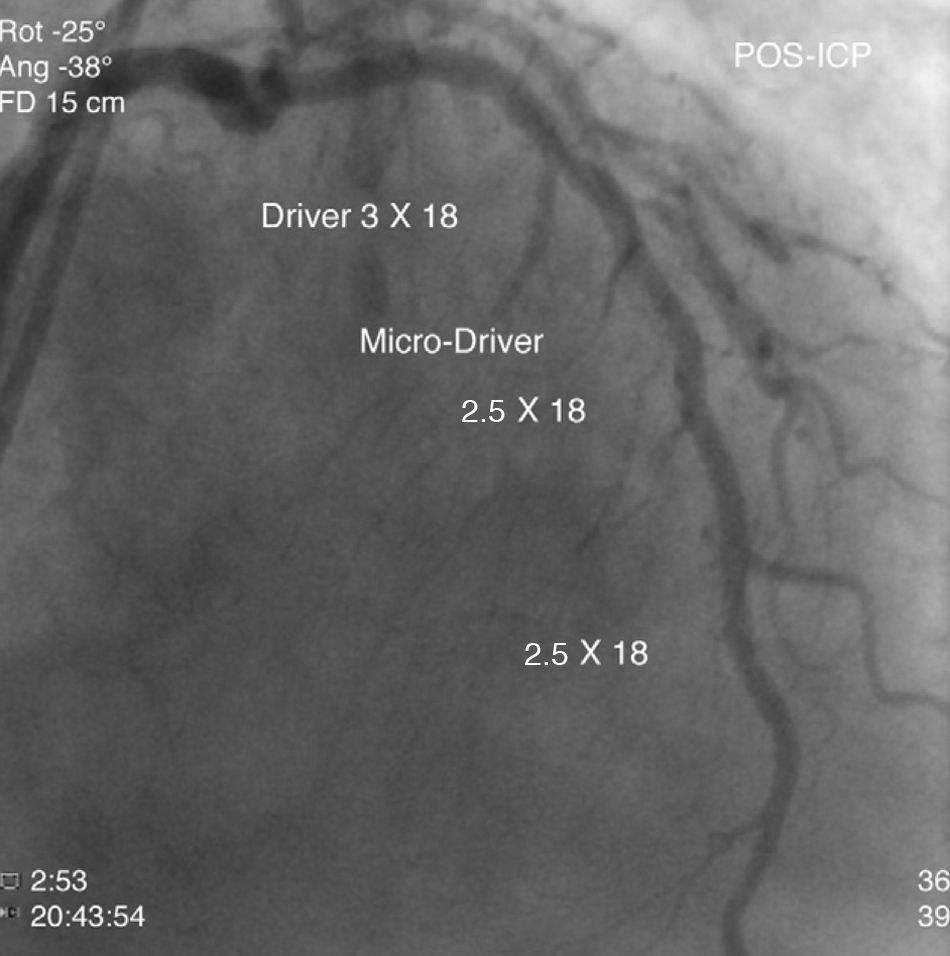
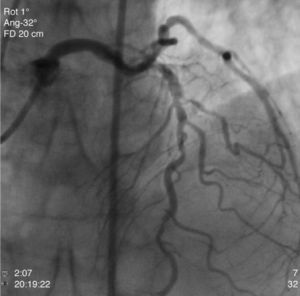
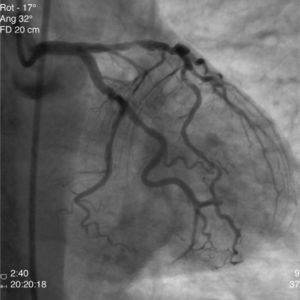
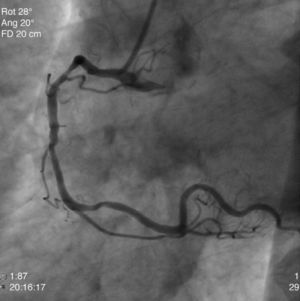
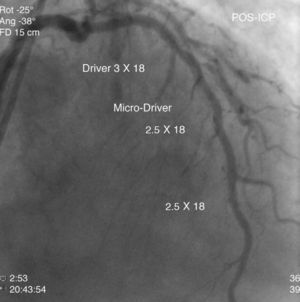
The patient was transferred to our hospital for direct angioplasty with a diagnosis of extensive ST-segment elevation acute coronary syndrome (ACS) of the anterior wall of 4h evolution. Coronary angiography revealed three-vessel disease with subocclusive stenosis of the proximal (culprit lesion) and distal segments of the left anterior descending artery (LAD), critical stenosis of the mid segment of the LAD, and significant stenoses of the right coronary and circumflex arteries. Three bare-metal stents were implanted in the LAD with a good result. There was no recurrence of angina during hospital stay and he remained hemodynamically and electrically stable and in Killip class 1. The ECG showed Q waves associated with ST-segment elevation and T-wave inversion in V1–V4, and laboratory tests showed peak troponin I>100ng/ml, CK-MB 105.6ng/ml and peak BNP 2490pg/ml.
He was transferred back to the first hospital with a diagnosis of myocardial infarction (MI) precipitated by taking zolmitriptan (Figs. 1–5).
The triptans are a well-established acute treatment for migraine.1 They are absorbed rapidly and onset of action typically occurs 30–60min after intake. They act as 5-HT1B/1D agonists, inducing constriction of the cerebral vessels that dilate during migraine attacks.2 They also inhibit secretion of vasoactive peptides and conduction of painful stimuli to the cerebral cortex.2 Although the therapeutic action of triptans occurs mainly through constriction of cerebral vessels, they have also been shown to have vasoconstrictive effects on the coronary, pulmonary, renal and systemic vasculature.3 The use of triptans is associated with chest pain in 1–7% of cases; the pain is mostly mild and transient and is not explained by ischemia.4 The incidence of MI in patients medicated with triptans is low, most reported cases being associated with sumatriptan,5,6 in patients with6,7 and without8 angiographically significant coronary disease. However, cases have been described of ACS after the use of other triptans, including zolmitriptan7 and frovatriptan.
We report a rare case of MI precipitated by taking zolmitriptan in a patient with high cardiovascular risk and angiographically significant coronary disease. According to a Medline search, this is the third such case in the literature. The first was reported by Mikhail et al. in 20049 and the second by Pacheco-Coronado et al. in 2009,10 but unlike our case, these reported MI in patients without cardiovascular risk factors and with angiographically normal coronary arteries. As suggested by Evans and Martin,11 we warn against using these drugs in patients with known ischemic heart disease (an absolute contraindication) and stress the need for cardiovascular assessment in individuals with risk factors for ischemic heart disease. It should also be borne in mind that ACS associated with triptan use can occur in individuals without vascular risk factors or significant coronary disease, and thus all patients using these drugs should be advised of the symptoms of myocardial ischemia and should be assessed rapidly and thoroughly if such symptoms occur.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Ribeiro H. Enfarte agudo do miocárdio após a toma de zolmitriptano. Rev Port Cardiol; 2012 doi:10.1016/j.repc.2011.12.005.