Hypercalcemia is a known cause of heart rhythm disorders, but it is rarely associated with ventricular arrhythmias. The authors present the case of a 53-year-old man with ischemic and alcoholic dilated cardiomyopathy and severely reduced ejection fraction, with a cardiac resynchronization therapy-cardioverter-defibrillator (ICD) device, who was admitted to the emergency department due to an electrical storm with multiple appropriate ICD shocks, refractory to antiarrhythmic therapy. Etiological investigation documented severe hypercalcemia secondary to previously undiagnosed primary hyperparathyroidism. The episodes of ventricular tachycardia only ceased after serum calcium levels were reduced.
A hipercalcemia é uma causa conhecida de perturbações do ritmo cardíaco, contudo, a sua associação a arritmias ventriculares é rara. Os autores apresentam o caso clínico de um doente de 53 anos de idade, com cardiomiopatia dilatada de etiologia isquémica e etanólica, e grave compromisso da função sistólica global, portador de sistema de ressincronização cardíaca (CRT) com cardioversor desfibrilhador (CDI), admitido no serviço de urgência por tempestade arrítmica, com múltiplos choques de CDI apropriados, refratária a terapêutica antiarrítmica. Na investigação etiológica foi documentada hipercalcemia grave secundária a hiperparatiroidismo primário até então desconhecido. Somente após redução da calcemia se observou cessação dos episódios de taquicardia ventricular.
A 53-year-old Caucasian man with several cardiovascular risk factors (hypertension and heavy smoking and alcohol consumption) had a history of non-ST-elevation acute coronary syndrome at the age of 49 years and severe disease of the mid and distal right coronary artery, which were treated by angioplasty and implantation of two bare-metal stents. Because of persistent severe left ventricular dysfunction (biplane left ventricular ejection fraction of 15%) even on optimal medical therapy, an atrioventricular cardiac resynchronization therapy (CRT)/implantable cardioverter-defibrillator (ICD) system was implanted.
Three years later, he experienced an episode of monomorphic ventricular tachycardia (VT) with a pattern of complete right bundle branch block and superior axis prompting an ICD shock followed by chest pain and elevation of troponin I levels (peak 1.69 mg/l, cutoff <0.07). He underwent repeat coronary angiography that showed in-stent occlusion, treated by angioplasty and placement of three drug-eluting stents. Among the patient's other known personal history were smoking-related chronic obstructive pulmonary disease, erosive gastritis and peptic ulcer. He was medicated as an outpatient with losartan, bisoprolol, spirolactone, furosemide, aspirin, clopidogrel, amiodarone, simvastatin and pantograph. He remained in New York Heart Association class II from that time.
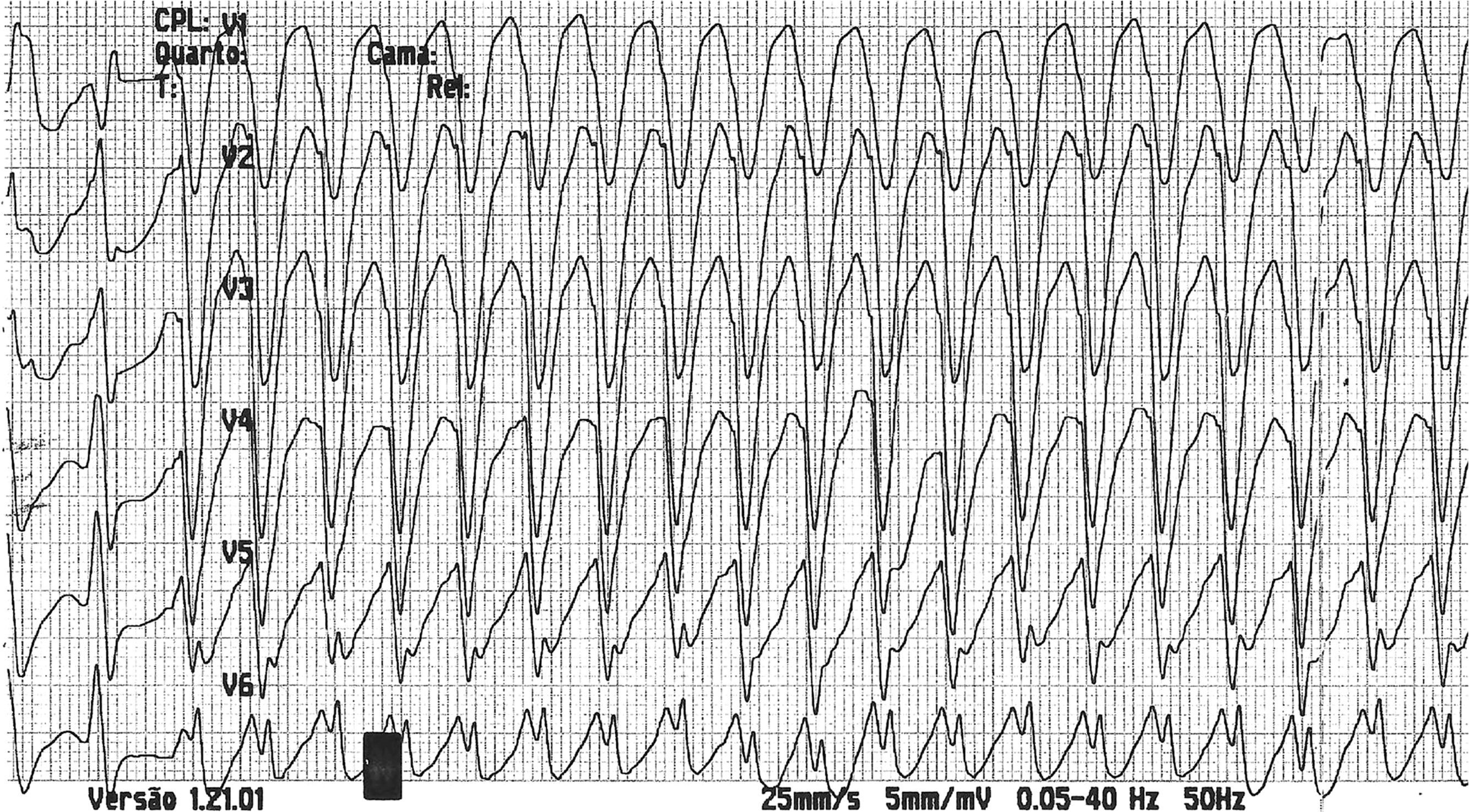
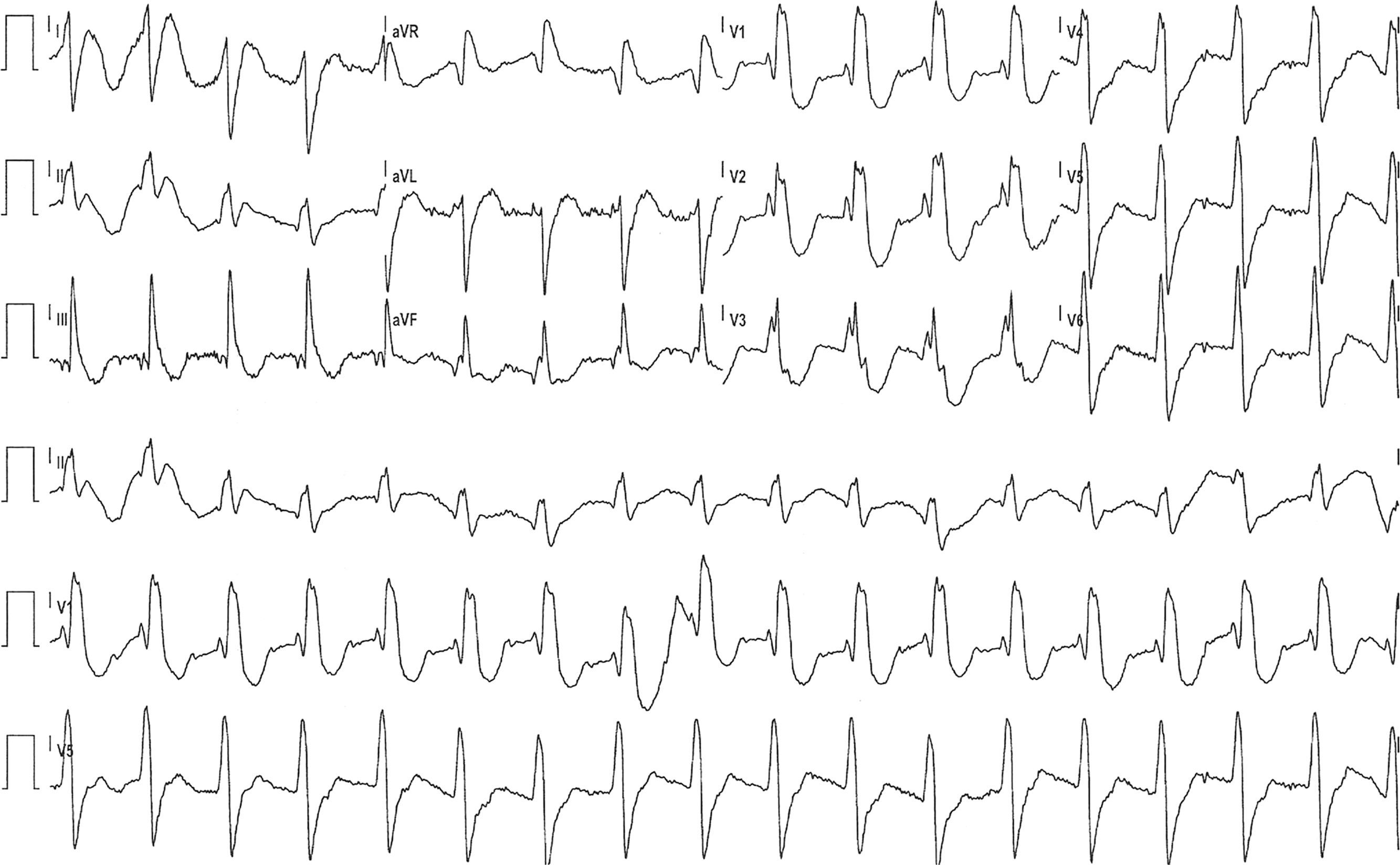
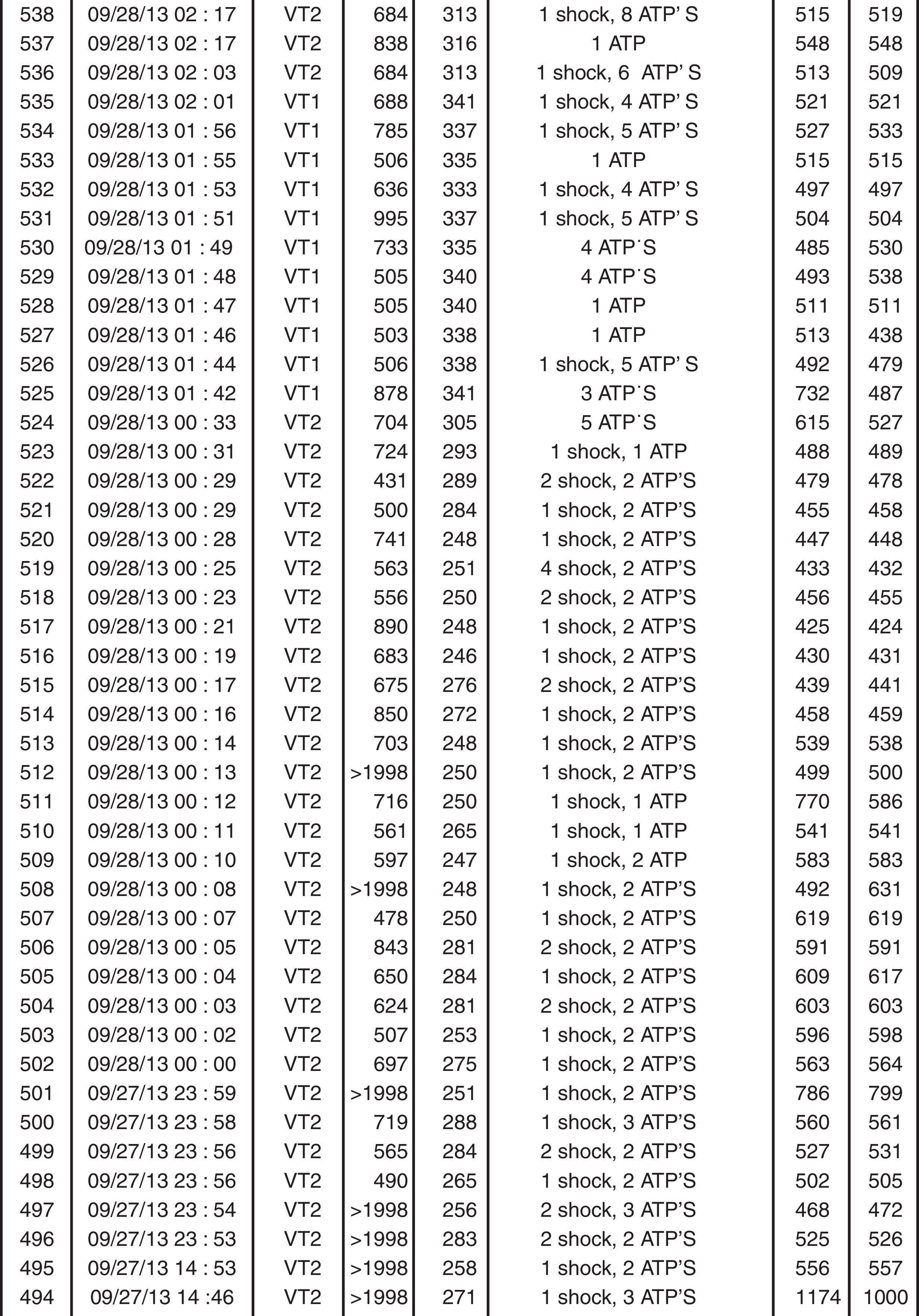
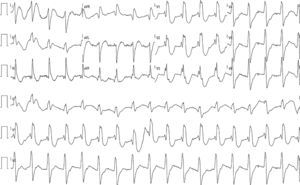
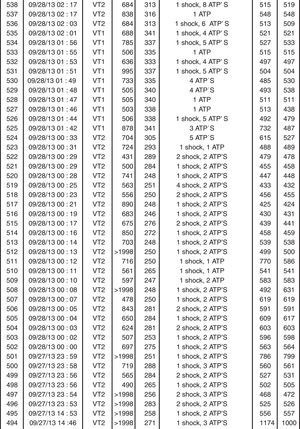
The patient was readmitted to our hospital one year after the previous hospitalization due to multiple ICD shocks, preceded by syncope, with transient recovery of consciousness following each shock, beginning 24 hours previously and increasing in frequency in the hour before admission. He had discontinued his medication in the previous five days due to financial difficulties, since when he had complained of anorexia, asthenia, constipation and dizziness. On admission he was conscious, oriented, and sweating but with no signs of peripheral hypoperfusion. The electrocardiogram (ECG) documented monomorphic VT with complete left bundle branch block and a rate of 300 bpm (Figure 1). A 300-mg bolus of amiodarone was administered followed by intravenous (IV) perfusion at 4 μg/kg/min, but recurrence of VT (with constant morphology on telemetry) was observed; two boluses of IV lidocaine (total 150 mg) were administered but also had no effect. The patient received multiple ICD shocks immediately after arrival at the hospital, resulting in the rapid exhaustion of the generator before the cardiologist was able to inhibit the therapies with the aid of a magnet. During periods of sinus rhythm, the ECG documented QRS with complete right bundle branch block, PR interval at the upper normal level, and corrected QT interval of 405 ms, with no signs of acute ischemia (Figure 2). Transthoracic echocardiography showed a severely dilated left ventricle, with diffuse hypokinesia and severely impaired global systolic function. Interrogation of the CRT-ICD revealed various episodes of VT in the previous 24 hours, with a total of 48 appropriate shocks (Figure 3), and the generator at the end of battery life. Given the patient's worsening heart failure and persistence of the arrhythmic storm, he was intubated and given propofol and midazolam for sedation and analgesia. Laboratory tests at admission showed acute kidney injury (Acute Kidney Injury Network stage 1) with creatinine 1.5 mg/dl and urea 101 mg/dl; mild hypokalemia (potassium 3.0 mmol/l, normal range 3.5-5.1); severe hypercalcemia (calcium 18.5 mg/dl, normal range 8.6-10; Ca2+ 2.46 mmol/l, normal range 1.13-1.32); and slight elevation of troponin I (peak 1.5 mg/dl, cutoff <0.07). The hypokalemia was immediately corrected.
Since the patient's monomorphic VT was not associated with pain or anginal equivalents, and there were no signs of acute ischemia on the baseline ECG, the cause of the arrhythmia was assumed to be the presence of severe hypercalcemia. With the support of internal medicine and endocrinology specialists, pharmacological measures were instituted to correct hypercalcemia with pamidronate, zolendronic acid, furosemide and hydrocortisone. However, since the hypercalcemia was severe and refractory to the above treatment, it was decided to begin continuous venovenous hemodiafiltration (CVVHDF); this resulted in progressive reduction in the frequency and duration of TV episodes, which were no longer sustained and ceased completely when calcemia reached 15.8 mg/dl. The patient was extubated and CVVHDF was discontinued after 48 hours, but furosemide (60 mg/day) was continued to maintain normocalcemia. The possibility of ablation to treat the VT was discussed but was not performed since the arrhythmia did not recur after correction of hypercalcemia.
Etiological investigation of hypercalcemia showed marked elevation of parathyroid hormone (PTH) (1020 pg/ml, normal range 14-72), and normal albuminemia (3.5 g/dl, normal range 3.2-4.9), suggesting primary hyperparathyroidism (PHPT). Cervical ultrasound revealed a well-defined hypoechogenic homogeneous solid mass topographically slightly posterior and inferior to the left lobe of the thyroid gland suggestive of a parathyroid adenoma, and scintigraphy of the parathyroid glands documented a functional lesion of the left inferior parathyroid. These findings did not exclude the possibility of a malignant parathyroid tumor. Left inferior parathyroidectomy was proposed to the patient, who accepted, and the procedure was performed without complications.
Following parathyroidectomy the patient's calcemia and PTH levels normalized and so medical therapy for this purpose was discontinued. The pathology report confirmed parathyroid adenoma. The fact that there was no hypocalcemia or need for calcium therapy suggests that the hyperparathyroidism was recent or a worsening of previously less severe PHPT. Baseline endocrinological assessment at admission also showed elevated levels of gastrin (2130 pg/ml, normal range 13-115), chromogranin A (143.5 mmol/l, cutoff <6), and urinary metanephrines and hydroxyindolacetic acid, although these elevations were less marked and were absent on the second urine tests during hospital stay, thereby excluding pheochromocytoma. Endoscopy of the upper digestive tract revealed an ulcer in the lesser curvature of the stomach, compatible with chronic gastritis, and screening for Helicobacter pylori was positive. The presence of PHPT, previous history of gastric ulcer, and hypergastrinemia raised the possibility of type 1 multiple endocrine neoplasia, but the patient refused to undergo an octreotide scan to confirm this diagnosis.
After 24 days of hospitalization, following a favorable clinical course, the patient was discharged under optimal medical therapy for heart failure. He is being followed in the cardiology outpatient clinic and has had no new events to date.
DiscussionHypercalcemia is associated with cardiac rhythm disturbances, most often prolongation of the PR segment and the QRS interval and hence shortening of the QT interval,1 which is usually associated with bradycardia rather than tachycardia. The clinical situation reported here is thus most unusual. Only six cases of PHPT and hypercalcemia associated with ventricular arrhythmias have been described in the literature, the first of which, by Chaieb et al.,2 published in 1988, described a case of hyperparathyroidism and ventricular bigeminy. In 2000 Chang et al. reported the case of a woman without structural heart disease with PHPT and spontaneous sustained monomorphic VT, reproducible in electrophysiological study only following injection of calcium gluconate and terminated by verapamil.3 In 1993, 2003 and 2004 Kearney et al.,4 Kolb et al.5 and Kiewiet et al.,6 respectively, reported cases of polymorphic VT degenerating into ventricular fibrillation. The second case reported to date of monomorphic VT was published in 2004 by Occhetta et al.7
The mechanism by which hypercalcemia can trigger ventricular arrhythmias is the subject of debate. Proposed causes include early4 or late1 afterdepolarizations or reentry circuits secondary to shortening of the absolute refractory period.8 In PHPT, PTH itself, as well as hypercalcemia, has arrhythmogenic potential, which may explain the occurrence of ventricular arrhythmias with lower calcium levels than in the case reported. Studies in rat hearts show that PTH has direct positive inotropic and chronotropic effects, partly mediated by calcium inflow into myocardial cells.9 The combination of reduced conduction speed of the cardiac impulse and shortening of the refractory period thus makes reentrant mechanisms, and hence ventricular arrhythmias, more likely. It has been suggested that the presence of heart disease may contribute to the development of arrhythmic events in patients with hypercalcemia, although this relationship has yet to be confirmed.6
In the case presented, the presence of alcohol- and ischemia-related dilated cardiomyopathy, with transmural scar and impaired global systolic function, together with the patient's failure to take his medication and hypokalemia (even though mild), may have contributed to his VT. However, PHPT and resulting hypercalcemia was the most important factor in this situation, acting as the primary trigger, given that the VT episodes only ceased after significant reduction in calcemia, with restoration of normocalcemia and no recurrence of VT after parathyroidectomy. Nevertheless, the possibility cannot be excluded that high PTH levels also contributed to the patient's condition.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Guimarães T, Nobre Menezes M, Cruz D, do Vale S, Bordalo A, Veiga A, et al. Crise hipercalcémica e hiperparatiroidismo primário: causa de tempestade arrítmica invulgar. Rev Port Cardiol. 2017;36:959.e1–959.e5.