Increased matrix metalloproteinase-9 (MMP-9) levels in ST-elevation myocardial infarction (STEMI) are well established; however, existing data on MMP-9 values as a prognostic marker after STEMI are limited and have been conflicting.
ObjectiveThis study aimed to assess the clinical significance of MMP-9 in predicting two-year adverse cardiovascular events in patients who underwent primary percutaneous coronary intervention (PCI) after STEMI.
MethodsIn this prospective study, 204 patients with STEMI undergoing PCI were included. Participants were classified as high MMP-9 (n=102) or low MMP-9 (n=102) based on a cutoff of 12.92 ng/ml. Both groups were assessed at one and two years after STEMI.
ResultsHigher cardiovascular mortality at one year was observed in the high MMP-9 group (13.7% vs. 4.9% in the low MMP-9 group, p=0.03). When the follow-up period was extended to two years, the difference in cardiovascular mortality between the groups was more significant (17.6% vs. 4.9%, p=0.004). There was no significant difference at one-year follow-up in rates of advanced heart failure, however at the end of the second year, advanced heart failure was more prevalent in the high MMP-9 group (16.7% vs. 5.9%, p=0.015). After adjustment for potential confounders, a high MMP-9 value had 3.5-fold higher odds for cardiovascular mortality at two-year follow-up than low MMP-9.
ConclusionThese results suggest that high MMP-9 levels are a strong predictor of cardiovascular mortality and advanced heart failure at two-year follow-up in STEMI patients.
A elevação das metaloproteinases de matriz 9 (MPM-9) no enfarte do miocárdio com supradesnivelamento do segmento ST (EAMCSST) está bem estabelecida; no entanto, os dados existentes relativos aos valores de MPM-9 como marcadores de prognóstico após o EAMCSST são limitados e contraditórios.
ObjetivoAvaliar o significado prognóstico das MPM-9 em eventos cardiovasculares a dois anos nos doentes submetidos a intervenção coronária percutânea primária (ICP) após EAMCSST.
MétodosNeste estudo prospetivo, foram incluídos 204 doentes com EAMCSST submetidos a ICP. Os participantes foram agrupados no grupo de MPM-9 elevadas (n=102) ou no grupo de MPM-9 baixas (n=102) com base no valor MPM-9 de 12,92 ng/ml. Ambos os grupos foram avaliados a um e a dois anos após EAMCSST.
ResultadosA mortalidade cardiovascular mais elevada a um ano foi observada no grupo de MPM-9 elevadas (13,7% versus 4,9% no grupo com valores baixos, p=0,03). Quando o período de seguimento foi prolongado a dois anos, a diferença na mortalidade cardiovascular foi ainda superior (17,6% versus 4,9%, p=0,004). Não houve diferenças significativas no seguimento a um ano relativamente à insuficiência cardíaca avançada, mas no fim do segundo ano a insuficiência cardíaca avançada foi superior no grupo com MPM-9 elevadas (16,7% versus 5,9%, p=0,015). Após o ajuste para potenciais fatores confundentes, a presença de MPM-9 elevada aumentou 3,5 vezes a probabilidade de mortalidade cardiovascular no segundo ano de seguimento.
ConclusãoNíveis elevados de MPM-9 constituem um importante fator preditor da mortalidade cardiovascular e da insuficiência cardíaca avançada num período de dois anos após EAMCSST.
Acute coronary syndrome (ACS) remains one of the most important causes of mortality, despite advances in diagnosis and treatment.1 It is therefore important to perform risk assessment in ACS patients to improve long-term survival.
Matrix metalloproteinases (MMPs), a component of the extracellular matrix, play a role in tissue remodeling in both physiological and pathophysiological processes.2 Dysregulation of MMP expression contributes to tissue proteolysis and thus to thinning and rupturing of the atherosclerotic plaque, which is one of the main scenarios for the occurrence of acute coronary syndrome.3 It has been shown that MMP-9 levels increase significantly in various vascular conditions, especially acute myocardial infarction (AMI).4
Many indicators of adverse prognosis after AMI exist, some of which can be used not only for prognostic assessment but also for early diagnosis, such as creatinine kinase and troponins, which indicate cardiomyocyte necrosis.5–7 Previous studies showed that inflammation markers like C-reactive protein and serum amyloid A can be used for risk determination in post-AMI patients.8
It is thought that MMP-9 may play a role not only in the disease process but also in predicting adverse events in patients with coronary artery disease (CAD).9,10 MMP-9 may affect myocardial fibrosis via transforming growth factor beta signaling-induced periostin and connective tissue growth factor expression.11 In addition, MMP-9 was found to be three times higher in the infarct site in post-AMI patients, suggesting that MMP-9 is an important indicator for cardiac remodeling and prognosis in patients with AMI.12 However, existing data on the specific impact of baseline plasma MMP-9 levels and its utility as a prognostic marker after AMI are limited and have been conflicting.13–15 This study was designed to determine the clinical value of MMP-9 in predicting two-year adverse cardiovascular events in ST-elevation myocardial infarction (STEMI) patients who underwent primary percutaneous coronary intervention (PCI).
MethodsStudy populationA total of 204 consecutive patients who were admitted to a large-volume center with a diagnosis of STEMI and who underwent primary PCI between January 2010 and December 2011 were included in this prospective observational study. STEMI patients were defined as those with typical chest discomfort, or other ischemic symptoms, who developed new ST-segment elevation in two or more contiguous leads or new bundle branch block with ischemic repolarization pattern on the standard 12-lead electrocardiogram (ECG). All primary PCI procedures were performed by operators who carried out more than 100 PCIs/year (>3000 PCIs/year in the center as a whole). The minimum number of patients who needed to be included for an effect size of 0.2 and 90% power was calculated and 279 STEMI patients were included in the study. Some patients were subsequently excluded due to problems with follow-up. All patients were scheduled to be referred to the cardiology clinic at the end of the third month after discharge to undergo ECG, to determine the presence of cardiac symptoms and to check patient adherence to medical treatment, but 41 patients did not attend the three-month follow-up and were excluded from the study. A further eight patients who discontinued medical therapy or took their medications irregularly with no medical reason were also excluded. During the two-year follow-up, 26 patients who could not be interviewed and whose hospital records were not available to assess secondary outcomes were excluded from the study. The study population thus decreased to 204 patients. Since the statistical power of this number of 204 patients was satisfactory with the power for the final sample size calculated at 80%, we decided not to recruit additional patients. Sample size was calculated using the G*Power 3.9.1.2 statistical package and was also valid for other statistical tests used in the study. Written informed consent was obtained from all patients in accordance with the requirements of the Declaration of Helsinki, and approval was obtained for the study from our university's ethics committee.
Analysis of patient dataData on patient demographic parameters, past medical history and medical therapies were collected. Patients were classified according to the presence of risk factors and their past medical history. Smoking status was determined according to patients’ statements. Hypertension was defined as systolic blood pressure >140 mmHg and diastolic blood pressure >90 mmHg or having used antihypertensive drugs for longer than two weeks. In accordance with the European guidelines, the presence of hypercholesterolemia was determined according to cardiovascular risk and low-density lipoprotein cholesterol level or the use of statins. Patients who were prescribed medical therapy because of fasting blood glucose >126 mg/dl, impaired fasting glucose or impaired glucose tolerance were defined as having diabetes.
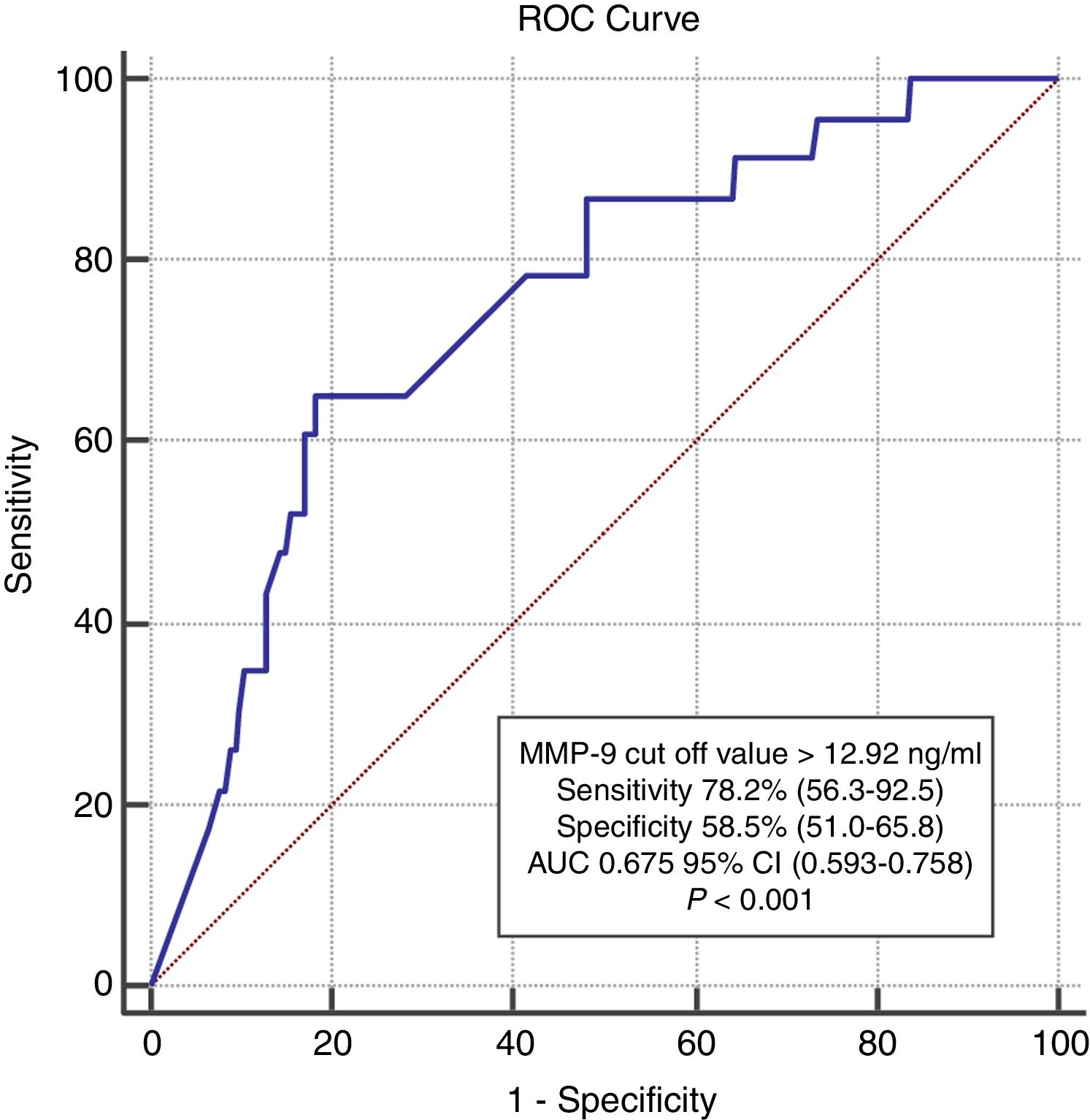
Five ml of peripheral venous blood was collected from all patients into EDTA-coated vacutainer tubes at the time of hospital admission, after which plasma samples were immediately separated by centrifugation and stored at -70°C. An enzyme-linked immunosorbent assay (ELISA) (Wuhan EIAab Science Col. Ltd., China) was used to determine MMP-9 levels, and its performance was evaluated using an Epoch microplate spectrophotometer (Biotek Instruments Inc., USA) at a sensitivity of 0.04 ng. The study population was divided into two groups, high and low MMP-9 (n=102 in each), based on individual plasma MMP-9 concentrations, using 12.92 ng/ml as the threshold value, which was the cut-off point with the highest combination of sensitivity and specificity calculated using receiver operating characteristic (ROC) analysis.
AMI type was identified for each patient immediately after emergency room admission by analyzing their 12-lead ECG. The biplane Simpson method was used to calculate left ventricular ejection fraction (LVEF) by transthoracic echocardiography (Vivid S5, 3S-RS probe, GE Healthcare, WI, USA) 24-72 hours after revascularization, as well as three months after discharge.16
Coronary intervention procedureAll participants received chewable 100 or 300 mg aspirin (according to previous use) and clopidogrel (600 mg loading dose) before coronary angiography. Heparin (100 IU/kg) was administered after the decision was made to perform the coronary intervention. After angioplasty, all patients were admitted to the coronary care unit, where routine antithrombotic therapy was given in the form of a daily dose of 100 mg aspirin and 75 mg clopidogrel. Routine enoxaparin was not given after a successful percutaneous procedure. Enoxaparin was given at prophylactic doses in immobilized patients, and at therapeutic doses in patients with Thrombolysis in Myocardial Infarction (TIMI) 0-1 flow grade after the procedure and in patients with atrial fibrillation. Patients’ angiographic data were obtained from catheterization laboratory records. Emergency coronary angiography and angioplasty were performed through femoral access. The artery that was assumed to be the non-infarct-related artery was injected first. Blood flow in the infarct-related artery (IRA) was calculated using the TIMI classification.17 Primary angioplasty was performed only for IRA occlusion (total or partial). Intervention success was defined as reduction of IRA obstruction and stenosis to ≤30% with TIMI 3 flow after coronary intervention. Glycoprotein IIb/IIIa inhibitors were administered as bailout if there was evidence of no-reflow or a thrombotic complication.
DefinitionsReperfusion time was measured as the time from symptom onset to balloon inflation to achieve coronary reperfusion. Door-to-balloon time was defined as the time between emergency room admission and balloon inflation. Killip class was recorded for each patient.18 Patients in New York Heart Association functional class ≥III despite optimal medical treatment and with LVEF ≤35% (calculated three months after discharge) were defined as having advanced heart failure. Cardiovascular mortality was defined as sudden death due to AMI, arrhythmia or heart failure. Target vessel revascularization was described as the need for PCI or coronary artery bypass graft surgery due to severe restenosis or reocclusion of the IRA. Reinfarction was defined according to the World Health Organization criteria.19
Follow-upPatients’ data during follow-up visits were obtained from hospital records or by interviewing patients, their families or their physicians in person or by telephone. Patients with suspect clinical data were excluded from the study. The primary clinical outcome was cardiovascular (CV) mortality, and secondary clinical outcomes were defined separately as target vessel revascularization (TVR), non-fatal reinfarction, stroke and advanced heart failure. Although some data are obtained from national databases on patient deaths, patient interviews were mandatory to detect the presence of TVR, stroke, heart failure, or non-fatal AMI as secondary outcomes. For this reason, patients who could not be contacted by telephone or other communication tools during the two-year follow-up were excluded from the study.
Statistical analysisThe statistical analysis was performed using SPSS for Windows, version 18.0 (SPSS Inc., Chicago, IL, USA). Normality of distribution was determined using visual and analytical methods. Descriptive analyses are presented as means and standard deviations for variables with normal distribution and as median and interquartile range for non-normal distribution. Categorical variables are expressed as numbers and percentages. Study groups were compared using the unpaired Student's t test for continuous variables with a normal distribution and the Mann-Whitney U test for continuous variables with a non-normal distribution. Categorical data were compared with the chi-square test. ROC curves were drawn for the ability of MMP-9 plasma levels to predict cardiovascular mortality. Youden's index was used to derive the best cut-off value for plasma MMP-9 concentrations. On the basis of this value, patients were divided into two groups, high and low MMP-9. A cumulative survival curve for two-year cardiovascular mortality was obtained using the Kaplan-Meier method, with differences assessed by log-rank tests between the two groups. Backward stepwise multivariate logistic regression including variables with p<0.1 on univariate analysis was carried out to identify independent predictors of two-year cardiovascular mortality. A p-value of <0.05 was considered statistically significant.
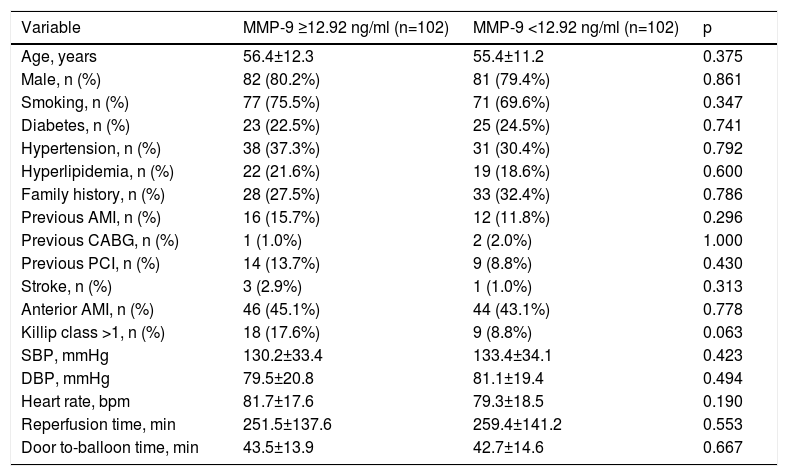
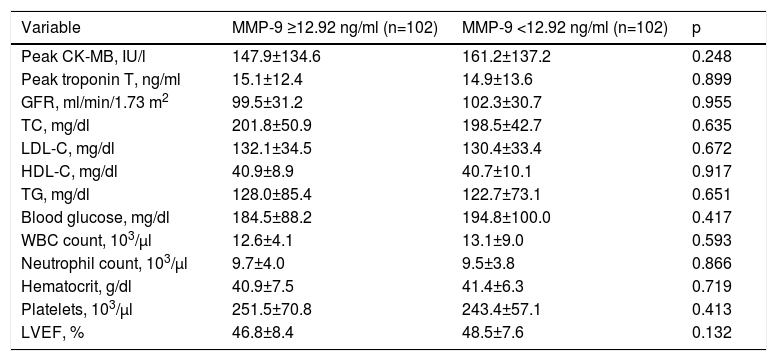
ResultsAfter removing cases based on the exclusion criteria, a total of 204 patients remained in the study. There were no differences between the high and low MMP-9 groups in terms of baseline characteristics or laboratory values (Tables 1 and 2), and data on coronary intervention were similar between the study groups. Moreover, cardiac medical therapy after discharge was prescribed in around 90% of both groups, without statistical difference (Table 3). Nonetheless, cardiogenic shock (31.4% vs. 1.8%, p<0.001), need for inotropic therapy (34.3% vs. 3.0%, p<0.001) and intra-aortic balloon pump (IABP) use (14.3% vs. 1.8%, p=0.003) were more frequent in patients with no-reflow compared to patients with reflow.
Baseline characteristics of the study population.
| Variable | MMP-9 ≥12.92 ng/ml (n=102) | MMP-9 <12.92 ng/ml (n=102) | p |
|---|---|---|---|
| Age, years | 56.4±12.3 | 55.4±11.2 | 0.375 |
| Male, n (%) | 82 (80.2%) | 81 (79.4%) | 0.861 |
| Smoking, n (%) | 77 (75.5%) | 71 (69.6%) | 0.347 |
| Diabetes, n (%) | 23 (22.5%) | 25 (24.5%) | 0.741 |
| Hypertension, n (%) | 38 (37.3%) | 31 (30.4%) | 0.792 |
| Hyperlipidemia, n (%) | 22 (21.6%) | 19 (18.6%) | 0.600 |
| Family history, n (%) | 28 (27.5%) | 33 (32.4%) | 0.786 |
| Previous AMI, n (%) | 16 (15.7%) | 12 (11.8%) | 0.296 |
| Previous CABG, n (%) | 1 (1.0%) | 2 (2.0%) | 1.000 |
| Previous PCI, n (%) | 14 (13.7%) | 9 (8.8%) | 0.430 |
| Stroke, n (%) | 3 (2.9%) | 1 (1.0%) | 0.313 |
| Anterior AMI, n (%) | 46 (45.1%) | 44 (43.1%) | 0.778 |
| Killip class >1, n (%) | 18 (17.6%) | 9 (8.8%) | 0.063 |
| SBP, mmHg | 130.2±33.4 | 133.4±34.1 | 0.423 |
| DBP, mmHg | 79.5±20.8 | 81.1±19.4 | 0.494 |
| Heart rate, bpm | 81.7±17.6 | 79.3±18.5 | 0.190 |
| Reperfusion time, min | 251.5±137.6 | 259.4±141.2 | 0.553 |
| Door to-balloon time, min | 43.5±13.9 | 42.7±14.6 | 0.667 |
AMI: acute myocardial infarction; CABG: coronary artery bypass grafting; DBP: diastolic blood pressure; MMP: matrix metalloproteinase; PCI: percutaneous coronary intervention; SBP: systolic blood pressure; SD: standard deviation.
Values are mean ± SD or n (%).
Baseline laboratory findings of the study population.
| Variable | MMP-9 ≥12.92 ng/ml (n=102) | MMP-9 <12.92 ng/ml (n=102) | p |
|---|---|---|---|
| Peak CK-MB, IU/l | 147.9±134.6 | 161.2±137.2 | 0.248 |
| Peak troponin T, ng/ml | 15.1±12.4 | 14.9±13.6 | 0.899 |
| GFR, ml/min/1.73 m2 | 99.5±31.2 | 102.3±30.7 | 0.955 |
| TC, mg/dl | 201.8±50.9 | 198.5±42.7 | 0.635 |
| LDL-C, mg/dl | 132.1±34.5 | 130.4±33.4 | 0.672 |
| HDL-C, mg/dl | 40.9±8.9 | 40.7±10.1 | 0.917 |
| TG, mg/dl | 128.0±85.4 | 122.7±73.1 | 0.651 |
| Blood glucose, mg/dl | 184.5±88.2 | 194.8±100.0 | 0.417 |
| WBC count, 103/μl | 12.6±4.1 | 13.1±9.0 | 0.593 |
| Neutrophil count, 103/μl | 9.7±4.0 | 9.5±3.8 | 0.866 |
| Hematocrit, g/dl | 40.9±7.5 | 41.4±6.3 | 0.719 |
| Platelets, 103/μl | 251.5±70.8 | 243.4±57.1 | 0.413 |
| LVEF, % | 46.8±8.4 | 48.5±7.6 | 0.132 |
CK-MB: creatine kinase myocardial band; GFR: glomerular filtration rate; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; LVEF: left ventricular ejection fraction; MMP: matrix metalloproteinase; SD: standard deviation; TC: total cholesterol; TG: triglycerides; WBC; white blood cell.
Values are mean ± SD or n (%).
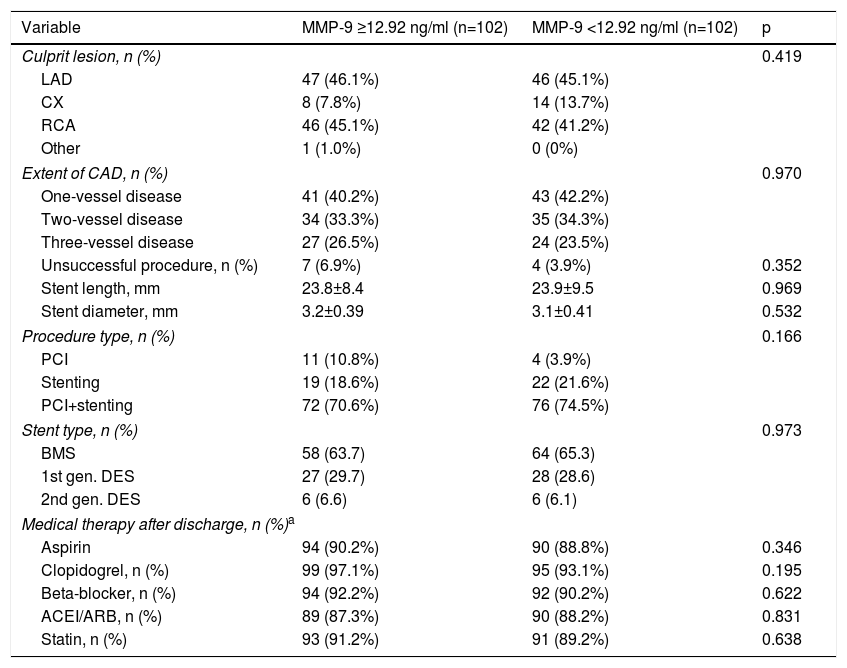
Angiographic and procedural characteristics and medical therapy in the study population.
| Variable | MMP-9 ≥12.92 ng/ml (n=102) | MMP-9 <12.92 ng/ml (n=102) | p |
|---|---|---|---|
| Culprit lesion, n (%) | 0.419 | ||
| LAD | 47 (46.1%) | 46 (45.1%) | |
| CX | 8 (7.8%) | 14 (13.7%) | |
| RCA | 46 (45.1%) | 42 (41.2%) | |
| Other | 1 (1.0%) | 0 (0%) | |
| Extent of CAD, n (%) | 0.970 | ||
| One-vessel disease | 41 (40.2%) | 43 (42.2%) | |
| Two-vessel disease | 34 (33.3%) | 35 (34.3%) | |
| Three-vessel disease | 27 (26.5%) | 24 (23.5%) | |
| Unsuccessful procedure, n (%) | 7 (6.9%) | 4 (3.9%) | 0.352 |
| Stent length, mm | 23.8±8.4 | 23.9±9.5 | 0.969 |
| Stent diameter, mm | 3.2±0.39 | 3.1±0.41 | 0.532 |
| Procedure type, n (%) | 0.166 | ||
| PCI | 11 (10.8%) | 4 (3.9%) | |
| Stenting | 19 (18.6%) | 22 (21.6%) | |
| PCI+stenting | 72 (70.6%) | 76 (74.5%) | |
| Stent type, n (%) | 0.973 | ||
| BMS | 58 (63.7) | 64 (65.3) | |
| 1st gen. DES | 27 (29.7) | 28 (28.6) | |
| 2nd gen. DES | 6 (6.6) | 6 (6.1) | |
| Medical therapy after discharge, n (%)a | |||
| Aspirin | 94 (90.2%) | 90 (88.8%) | 0.346 |
| Clopidogrel, n (%) | 99 (97.1%) | 95 (93.1%) | 0.195 |
| Beta-blocker, n (%) | 94 (92.2%) | 92 (90.2%) | 0.622 |
| ACEI/ARB, n (%) | 89 (87.3%) | 90 (88.2%) | 0.831 |
| Statin, n (%) | 93 (91.2%) | 91 (89.2%) | 0.638 |
ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; BMS: bare-metal stent; CAD: coronary artery disease; CX: circumflex; DES: drug-eluting stent; gen.: generation; LAD: left anterior descending; MMP: matrix metalloproteinase; PCI: percutaneous coronary intervention; RCA: right coronary artery; SD: standard deviation.
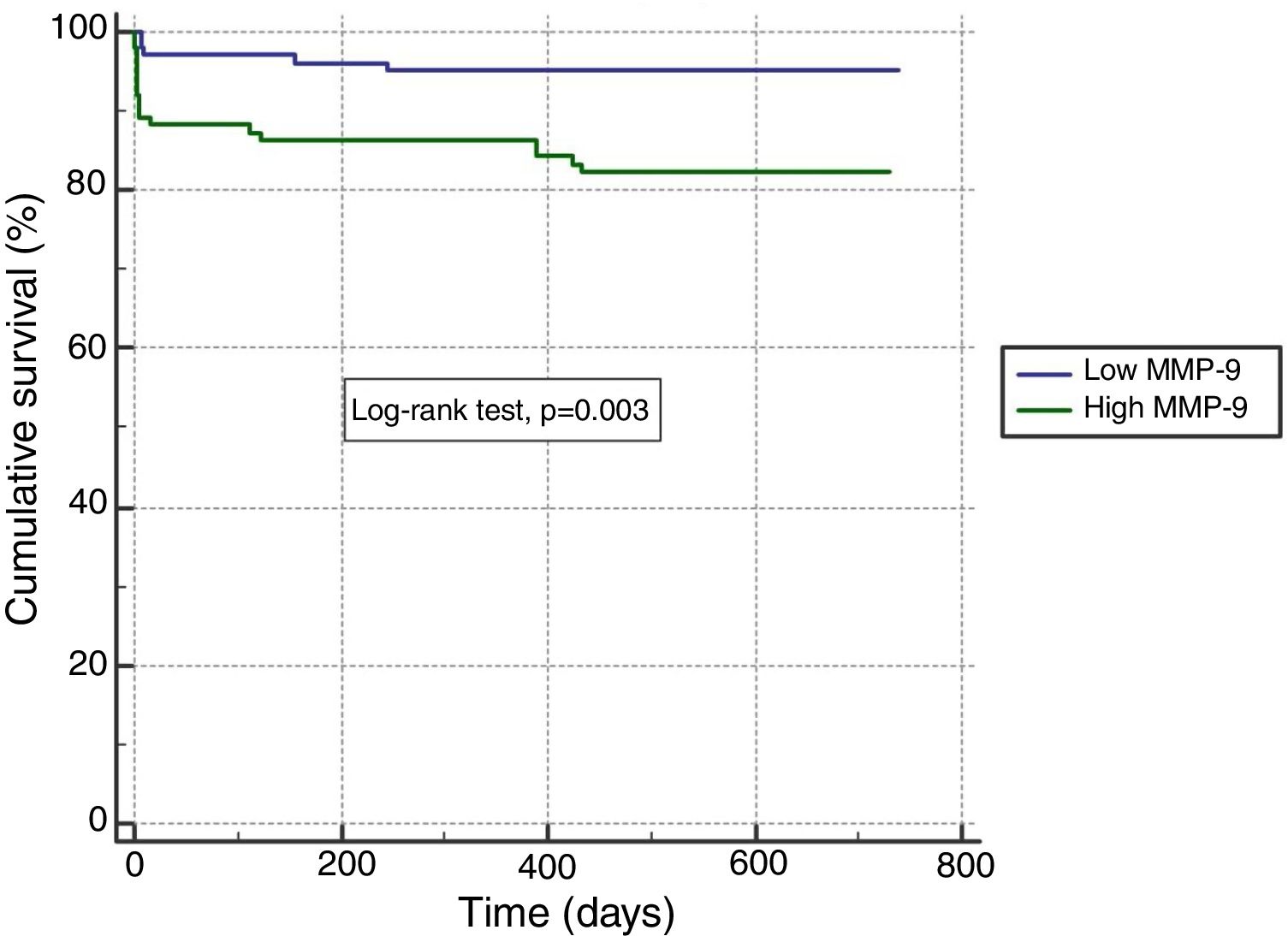
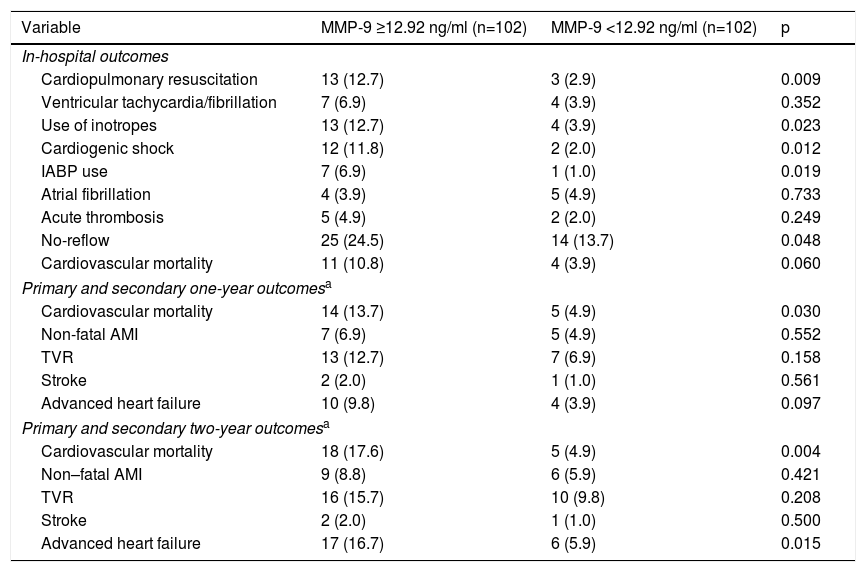
In our study, the cardiovascular mortality rate was 7.3% in-hospital, 9.3% at one-year follow-up and 11.2% at two-year follow-up. Although in-hospital cardiovascular mortality was higher in the high MMP-9 group than the low MMP-9 group, this was not statistically significant. However, the high MMP-9 group had a significantly higher incidence of cardiopulmonary resuscitation, use of inotropes, cardiogenic shock, IABP use and no-reflow phenomenon. Furthermore, cardiogenic shock (31.4% vs. 1.8%, p<0.001), need for inotropic therapy (34.3% vs. 3.0%, p<0.001) and IABP use (14.3% vs. 1.8%, p=0.003) were more frequent in patients with no-reflow compared to patients with reflow. When long-term outcomes were analyzed, one-year cardiovascular mortality was significantly higher in the high MMP-9 group (13.7% vs. 4.8% for the low MMP-9 group, p=0.030), unlike in-hospital outcomes (10.8% vs. 3.9%, p=0.060). Furthermore, when the follow-up period was extended to two years, the difference in cardiovascular mortality between the groups was also statistically significant (17.6% vs. 4.9%, p=0.004; Table 4), with more serious outcomes for the high MMP-9 group. In parallel with the above-mentioned results, we found that the high MMP-9 group had 3.5-fold higher odds for two-year cardiovascular mortality than the low MMP-9 group, independently of age and LVEF (Table 5).
In-hospital, one-year and two-year outcomes of the overall study population.
| Variable | MMP-9 ≥12.92 ng/ml (n=102) | MMP-9 <12.92 ng/ml (n=102) | p |
|---|---|---|---|
| In-hospital outcomes | |||
| Cardiopulmonary resuscitation | 13 (12.7) | 3 (2.9) | 0.009 |
| Ventricular tachycardia/fibrillation | 7 (6.9) | 4 (3.9) | 0.352 |
| Use of inotropes | 13 (12.7) | 4 (3.9) | 0.023 |
| Cardiogenic shock | 12 (11.8) | 2 (2.0) | 0.012 |
| IABP use | 7 (6.9) | 1 (1.0) | 0.019 |
| Atrial fibrillation | 4 (3.9) | 5 (4.9) | 0.733 |
| Acute thrombosis | 5 (4.9) | 2 (2.0) | 0.249 |
| No-reflow | 25 (24.5) | 14 (13.7) | 0.048 |
| Cardiovascular mortality | 11 (10.8) | 4 (3.9) | 0.060 |
| Primary and secondary one-year outcomesa | |||
| Cardiovascular mortality | 14 (13.7) | 5 (4.9) | 0.030 |
| Non-fatal AMI | 7 (6.9) | 5 (4.9) | 0.552 |
| TVR | 13 (12.7) | 7 (6.9) | 0.158 |
| Stroke | 2 (2.0) | 1 (1.0) | 0.561 |
| Advanced heart failure | 10 (9.8) | 4 (3.9) | 0.097 |
| Primary and secondary two-year outcomesa | |||
| Cardiovascular mortality | 18 (17.6) | 5 (4.9) | 0.004 |
| Non–fatal AMI | 9 (8.8) | 6 (5.9) | 0.421 |
| TVR | 16 (15.7) | 10 (9.8) | 0.208 |
| Stroke | 2 (2.0) | 1 (1.0) | 0.500 |
| Advanced heart failure | 17 (16.7) | 6 (5.9) | 0.015 |
Values are n (%). AMI: acute myocardial infarction; IABP: intra-aortic balloon pump; MMP: matrix metalloproteinase; TVR: target vessel revascularization.
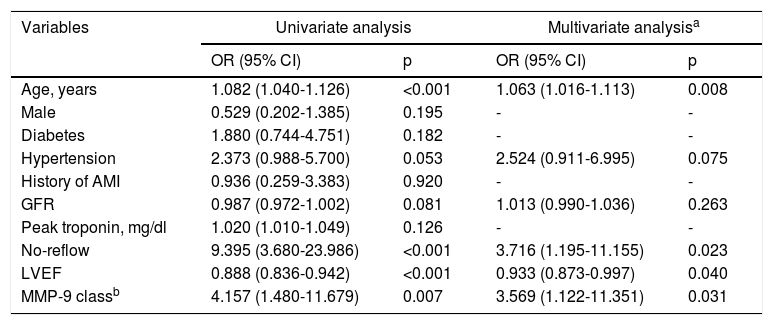
Multivariate logistic regression analysis for potential predictors of two-year cardiovascular mortality.
| Variables | Univariate analysis | Multivariate analysisa | ||
|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | |
| Age, years | 1.082 (1.040-1.126) | <0.001 | 1.063 (1.016-1.113) | 0.008 |
| Male | 0.529 (0.202-1.385) | 0.195 | - | - |
| Diabetes | 1.880 (0.744-4.751) | 0.182 | - | - |
| Hypertension | 2.373 (0.988-5.700) | 0.053 | 2.524 (0.911-6.995) | 0.075 |
| History of AMI | 0.936 (0.259-3.383) | 0.920 | - | - |
| GFR | 0.987 (0.972-1.002) | 0.081 | 1.013 (0.990-1.036) | 0.263 |
| Peak troponin, mg/dl | 1.020 (1.010-1.049) | 0.126 | - | - |
| No-reflow | 9.395 (3.680-23.986) | <0.001 | 3.716 (1.195-11.155) | 0.023 |
| LVEF | 0.888 (0.836-0.942) | <0.001 | 0.933 (0.873-0.997) | 0.040 |
| MMP-9 classb | 4.157 (1.480-11.679) | 0.007 | 3.569 (1.122-11.351) | 0.031 |
AMI: acute myocardial infarction; CI: confidence interval; GFR: glomerular filtration rate; LVEF: left ventricular ejection fraction; MMP: matrix metalloproteinase; OR: odds ratio.
From the secondary endpoints, advanced heart failure was compared between the groups in order to investigate the ability of MMP-9 to predict left ventricular remodeling. While there was no significant difference at the end of the first year (9.8% vs. 3.9%, p=0.098), there was a statistically significant higher prevalence of advanced heart failure in the high MMP-9 group at two-year follow-up (16.7% vs. 5.9%, p=0.015).
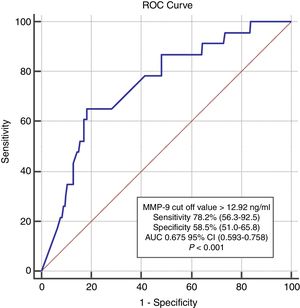
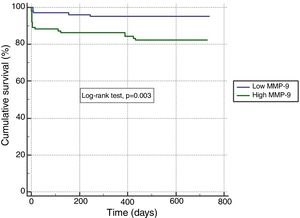
In ROC analysis, the area under the curve for MMP-9 ≥12.92 ng/ml was 0.675 for predicting two-year-cardiac mortality with 74.4% sensitivity and 55.6% specificity (Figure 1). The Kaplan-Meier survival plot for two-year CV mortality for the high and low MMP-9 groups is presented in Figure 2.
In our study, higher MMP-9 levels were associated with a marked increase in two-year cardiovascular mortality. Furthermore, as a secondary outcome, cases of advanced heart failure were significantly more frequent in the high MMP-9 group during two-year follow-up. Another notable aspect of the study was that, while there was no significant difference in in-hospital cardiovascular mortality between the groups, it was significantly different in long-term follow-up and was more marked as the follow-up period increased. Furthermore, it was noted that high MMP-9 was one of the independent predictors of two-year cardiovascular mortality (odds ratio [OR]: 3.5; 95% confidence interval [CI]: 1.12-11.35). Age, no-reflow phenomenon and LVEF were the other independent predictors of two-year cardiovascular mortality.
Levels of MMP-9, which is associated with plaque instability and rupture and with left ventricular remodeling due to its role in collagen-cleaving and degrading extracellular matrix proteins in plaque, have proved useful in predicting adverse cardiovascular events. Kaptoge et al.20 reported that high MMP-9 levels were associated with adverse cardiovascular events in a population-based study. In another prospective analysis, MMP-9 levels were high in patients with AMI, but were not identified as independent predictors due to their close relationship with other cardiovascular risk factors, such as C-reactive protein and smoking.15 Previous studies had established that serum MMP-9 levels could also be associated with both severity and progression of CAD.21,22 Furthermore, Blackenberg et al.23 demonstrated that plasma MMP-9 was a predictor of future CV mortality in patients with a history of CAD, independently of potential confounders. Although MMP-9| contributes to determining prognosis in population-based studies and individuals with coronary artery disease, there are conflicting data on predicting adverse CV events after AMI. For this reason, we felt the need to perform a study to clarify this situation.
As mentioned above, matrix metalloproteinases promote plaque instability and left ventricular remodeling, and in this study we set out to analyze the clinical responses to these two effects. Firstly, the role of MMP-9 in plaque instability was investigated through different parameters affecting CV mortality and morbidity. The effect of MMP-9 levels on plaque rupture was noted and the no-reflow phenomenon was seen more often in patients with ruptured plaques in previous studies.24,25 For this reason, in our study, the relation of MMP-9 levels with no-reflow was investigated, and no-reflow was statistically more frequent in the high MMP-9 group (p=0.048), as expected. Moreover, inadequate coronary flow may cause a domino effect, and no-reflow during the procedure may lead to cardiogenic shock or the need for IABP or inotropes, development of advanced heart failure and ultimately cardiovascular mortality. On the basis of this hypothesis, these consequences were analyzed separately in our study, which showed that the use of IABP (p=0.019), inotropic therapy (p=0.023) and cardiogenic shock (p=0.012) were more frequent in the high MMP-9 group. Furthermore, no-reflow was an independent predictor of two-year cardiovascular mortality (OR: 3.7; 95% CI: 1.19-11.15).
Many indicators of adverse prognosis after AMI exist, one of the most important of which is left ventricular remodeling, the second major effect of MMP-9. Infarcted and non-infarcted areas of the ventricle are affected by remodeling, which is caused by maladaptive changes to cellular and extracellular elements of the myocardium. MMP-9 has a role in regulating vascular tone and platelet aggregation, which are involved in adverse left ventricular remodeling.26 Especially following AMI, MMP levels are associated with LV function, as shown by echocardiography, establishing that they potentially affect LV remodeling. Kelly et al.27 showed that higher MMP-9 levels were associated not only with more severe impairment of LV function, but also with more extensive LV remodeling in the follow-up of post-AMI patients. In addition, Wagner et al.28 followed AMI patients treated with primary mechanical reperfusion for two years and found that MMP-9 levels were predictive of late-onset congestive heart failure after AMI. This is supported by other studies, which show that ACS patients with ruptured plaques that contain higher MMP levels compared to those without have larger infarct sizes, which causes more remodeling.29–31 Similarly to the above-mentioned studies, while there was no statistically significant difference in occurrence of advanced heart failure between the high and low MMP-9 groups at one-year follow-up in our study, there was a substantial increase in advanced heart failure correlating with follow-up duration in both groups, due to long-term LV remodeling progression (one-year follow-up, MMP-9 high vs. low group: 9.8% vs. 3.9%, p=0.097; two-year follow-up, MMP-9 high vs. low group: 16.7% vs. 5.9%, p=0.015). In other studies examining the relationship between MMP levels and remodeling, lower MMP-9 levels were seen in patients suffering from dilated cardiomyopathy, ischemic cardiomyopathy with LV assist device, or after cardiac resynchronization therapy.32 As a result, reverse ventricular remodeling in spite of medical treatment accompanied by decreased serum MMP-9 levels indirectly supports our finding that those with high MMP-9 serum levels may have a poor prognosis.
The other essential factors determining prognosis after ACS are reinfarction and revascularization. In this regard, Wang et al.33 studied MMP levels in post-AMI patients and found that increased plasma MMP-9 levels predicted future coronary revascularization after an 18-month follow-up, suggesting a critical role for MMP-9 in atherothrombosis in patients with AMI (hazard ratio: 2.72; 95% CI: 1.24-5.98; p=0.026). In our research, although non-fatal reinfarction and revascularization were more common in those with high MMP-9 levels, this was not statistically significant (p=0.421 and 0.208, respectively). Although we do not have precise data, it can be assumed that there were more fatal MIs in the high MMP-9 group. Besides, statistical significance might have been reached with a larger number of patients and longer follow-up periods. Consequently, we can speculate that circulating MMP-9 levels might have been related to severity of atherosclerosis and plaque rupture, which can result in adverse CV events in post-AMI patients.
There are some limitations to our study. First, this was a single-center analysis, which could lead to selection bias. Second, the number of patients was kept relatively low in order to establish a direct relationship between MMP-9 and adverse clinical outcome. Furthermore, our analysis included only MMP-9 levels. Thus, we cannot comment on other MMPs or tissue inhibitors, which is a large family of extracellular matrix turnover markers, assessment of which might have provided more information about prognosis. However, this study may be a stepping stone in achieving more accurate results by prompting studies on other members of the matrix metalloproteinase group and with more patients. Third, it is difficult to compare our results with other studies on this subject, due to differences in pre-analysis medical therapy, storage techniques and ELISA kits. Also, repeated measurements of MMP-9 were not performed, so changes during follow-up could not be determined. MMP-9 levels were analyzed in samples stored at -70°C, and so we cannot rule out the possibility of protein degradation; it has been demonstrated that storage at this temperature does not prevent the degradation of MMP-9 over time.34 However, it would not affect the differences in results found between the two study groups, since there would be similar degradation in all samples. Fourth, we investigated the predictive value of MMP-9 using a cut-off of 12.92 ng/ml, which was acquired from ROC analysis. Therefore, our results can be assessed only for our cohort, and this cut-off may differ from previous studies. This issue needs validation in other large populations. Nevertheless, this limitation is present in all studies with specific populations on the use of biomarkers in clinical settings. Finally, as the new antiplatelet agents ticagrelor and prasugrel were not covered by social security in our country, we gave clopidogrel to all patients in association with aspirin, so we are unable to determine the exact effect of MMP-9 in AMI patients who use the new antiplatelets. In addition, femoral access was preferred because at the time of recruitment our operators were not experienced in the radial procedure. This approach does not conform to current guidelines. Moreover, the rate of bare-metal stent use in our study was quite high. At the time of patient data collection, drug-eluting stents were not strongly recommended in the guidelines. Also, financial constraints in our country led to a high rate of use of bare-metal stents in that period. However, as shown in Table 3, there was no significant difference between stent types according to MMP-9 groups.
ConclusionIn summary, we determined that high MMP-9 levels were an important predictor of major adverse CV events, including CV mortality and advanced heart failure, in a two-year follow-up period in STEMI patients undergoing primary PCI. Although routine testing for MMP-9 is not recommended in the general population, MMP-9 measurement should be considered for STEMI patients to predict prognosis, and more frequent follow-up may be considered in patients with high MMP-9 levels. More comprehensive investigation is required to clarify this outcome.
FundingNo funding was received for this research.
Conflicts of interestThe authors have no conflicts of interest to declare.
We are grateful to the chest diseases, biostatistics and biochemistry departments of Mehmet Akif Ersoy Thoracic and Cardiovascular Surgery Center, Training and Research Hospital for the attention and time they dedicated to this study.