Proportionality of secondary mitral regurgitation (sMR) may be a key factor in deciding whether a patient may benefit from mitral intervention. The aim of this study was to evaluate the prognostic value of two different concepts of proportionality and assess their ability to improve MR stratification proposed by the American Society of Echocardiography (ASE) guidelines.
MethodsWe conducted a retrospective analysis in patients with reduced left ventricular ejection fraction (LVEF) (<50%) and at least mild sMR. Proportionality status was calculated using formulas proposed by a) Grayburn et al. – disproportionate sMR defined as EROALVEDV >0.14; b) Lopes et al. – disproportionate sMR whenever measured EROA>theoretical EROA (determined as 50%×LVEF×LVEDVMitralVTI). Primary endpoint was all-cause mortality.
ResultsA total of 572 patients (69±12 years; 76% male) were included. Mean LVEF was 33±9%, with a median left ventricular end-diastolic volume of 174 mL [136;220] and a median effective regurgitant orifice area of 14 mm2 [8;22]. During mean follow-up of 4.1±2.7 years, there were 254 deaths. There was considerable disagreement (p<0.001) between both formulas: of 96 patients with disproportionate sMR according to Lopes’ criteria, 46 (48%) were considered proportionate according to Grayburn's; and of 62 patients with disproportionate sMR according to Grayburn's, 12 (19%) were considered proportionate according to Lopes’ formula.
In multivariate analysis, only Lopes’ definition of disproportionate sMR maintained independent prognostic value (hazard ratio 1.5; 95% confidence interval 1.07–2.1, p=0.018) and improved the risk stratification of ASE sMR classification.
ConclusionOf the two formulas available to define disproportionate sMR, Lopes’ model emerged as the only one with independent prognostic value while improving the risk stratification proposed by the ASE guidelines.
A proporcionalidade da regurgitação mitral secundária (sMR) pode ser um fator chave na decisão de que doentes podem beneficiar de intervenção mitral. O objetivo deste estudo foi de avaliar o valor prognóstico de dois modelos de proporcionalidade e aferir a sua capacidade para melhorar a estratificação da regurgitação mitral proposta pelas guidelines da ASE.
MétodosRealizamos um estudo retrospetivo com doentes com LVEF reduzida (<50%) e pelo menos sMR ligeira. O status de proporcionalidade foi calculado usando as fórmulas propostas por: a) Grayburn et al. – sMR desproporcional definida por EROALVEDV >0.14; b) Lopes et al. – sMR desproporcional quando o EROA medido>EROA teórico (determinado por 50%×LVEF×LVEDVMitralVTI). O endpoint primário foi mortalidade por qualquer causa.
ResultadosForam incluídos 572 pacientes (69±12 anos; 76% sexo masculino). LVEF média foi de 33±9%, com um LVEDV mediano de 174 mL [136;220] e um EROA mediano de 14 mm2 [8;22]. Após um follow-up médio de 4,1±2,7 anos, ocorreram 254 mortes. Verificou-se marcada discordância (p<0,001) entre ambas as fórmulas: de entre 96 doentes com sMR desproporcional pelo modelo de Lopes, 46 (48%) foram considerados proporcionais pela fórmula de Grayburn; de entre os 62 doentes com sMR desproporcional pelo modelo de Grayburn, 12 (19%) foram considerados proporcionais pelo modelo de Lopes.
Em análise multivariável, apenas a definição de desproporcionalidade descrita por Lopes manteve valor prognóstico independente (HR 1,5; 95%CI 1,07–2,1, p=0,018) e melhorou a estratificação de risco pela classificação da sMR da ASE.
ConclusãoDe entre as duas fórmulas disponíveis para definição sMR desproporcional, apenas o modelo de Lopes demonstrou valor prognóstico independente e melhorou a estratificação de risco proposta pelas guidelines da ASE.
Secondary mitral regurgitation (sMR) most commonly results from abnormal left ventricular size, shape, or function.1,2 Manifestation in patients with reduced left ventricular ejection fraction (LVEF) is associated with worse prognosis.1,2 Two recent landmark randomized clinical trials (MITRA-FR and COAPT) assessing the role of transcatheter mitral edge-to-edge repair (TEER) in sMR showed conflicting results.3,4 While MITRA-FR failed to show any benefit from intervention, the COAPT trial revealed a lower rate of HF hospitalization and all-cause mortality. While several reasons may account for the discrepancies found, the concept of sMR disproportionality has been raised as an important one.
Grayburn et al. were the first to introduce the concept of MR disproportionality as an elegant way to interpret the severity of sMR in line with left ventricular (LV) dilation and dysfunction, assuming that sMR is hemodynamically significant when regurgitant fraction (RF) is at least 50%.5 The model proposed by these authors assumes different lines of proportionality (the slope varies according to LVEF) that relate effective regurgitant orifice area (EROA) or regurgitant volume (RVol) to the LV end diastolic volume (LVEDV). As such, patients with MR EROA or RVol below the line of proportionality are considered to have non-severe/proportionate sMR, while patients above the line have disproportionate sMR. However, the only EROALVEDV published cutoff by Grayburn et al. is 0.14, which is only valid for a regurgitant fraction (RF) of 50% and a LVEF of 30%.6 Consequently, the clinical applicability of this cutoff is seriously compromised whenever LVEF is different from 30%.
Recently, Lopes et al. built upon the originally conceived by Grayburn et al. and created a patient-individualized formula in which a patient-specific theoretical cutoff of EROA/RVol is established according to individual LVEDV and LVEF (assuming also that hemodynamically significant sMR occurs when RF is at least 50%).7 Therefore, the model by Lopes et al. has the advantage of being versatile and applicable to any patient. Whenever the Doppler measured EROA/RVol is superior to the established patient-specific theoretical cutoff, then sMR is considered disproportionate.
At its core, both authors theorize that mitral intervention can achieve better outcomes when applied to patients with disproportionate sMR. However, it remains to be established which is the best way to assess sMR proportionality. The aim of this study was to evaluate the prognostic value of both proportionality concepts and assess their ability to improve MR stratification on top of the American Society of Echocardiography (ASE) grading guidelines.8
MethodsStudy population and patient assessmentWe studied a single-center retrospective cohort of patients who underwent transthoracic echocardiography from 2010 to 2018 and were found to have at least mild sMR and reduced LVEF (defined as <50%). Patients had to be ambulatory and have been on guideline-directed heart failure (HF) therapy for at least three months before being included. Patients aged <18 years, hospitalized for decompensated HF in the previous three months, with at least moderate aortic valve disease, previous valve intervention, hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy, left ventricular non-compaction cardiomyopathy or renal transplant were excluded. The study protocol was reviewed and approved by the local ethics committee, which waived the need for informed consent.
Transthoracic echocardiographic examinations were conducted with GE-Healthcare (Chicago, IL, USA) equipment. Data were retrieved from EchoPAC software (GE-Healthcare) for retrospective evaluation. Two-dimensional, M-mode and Doppler measurements (including MR quantification) were used based on the criteria of the ASE.
During follow-up, patients had regular medical visits. Therapy adjustments were left at the discretion of the attending physician. The primary endpoint was all-cause mortality.
Proportionality evaluationTwo frameworks for the evaluation of MR disproportionality were assessed in this cohort.
According to Lopes et al., proportionality was determined based on calculation of an individualized theoretical EROA cutoff determined by the formula: 50%×LVEF×LVEDVMitralVTI.7 EROA measured using the PISA method was then compared with this theoretical cutoff.7 Briefly, if measured EROA>individualized theoretical EROA, the patient was considered to have disproportionate sMR.7 If not, sMR was considered proportionate/non-severe.7
In order to determine disproportionality according to Grayburn et al., EROALVEDV ratio was calculated.5 The proportionality cutoff used was 0.14, according to published data (line of proportionality).6 Even though this cutoff was established specifically for patients with a LVEF of 30% at a RF of 50%, we considered that a ratio >0.14 identified disproportionate sMR, whereas a ratio ≤0.14 was categorized as non-severe/proportionate.
Mitral regurgitation severity was classified according to the EROA cutoffs proposed by the ASE classification to allow meaningful comparisons between the two above mentioned formulas of sMR proportionality (grade I [mild] if EROA<0.2 cm2; grade II [moderate] if EROA 0.2-0.29 cm2; grade III [moderate-severe] if EROA 0.3-0.39 cm2; and grade IV [severe] if EROA ≥0.4 cm2).8
Statistical analysisCategorical variables are presented as frequencies and percentages, and continuous variables as mean and standard deviation, or median and interquartile range for variables with skewed distributions. Differences among groups were assessed using Pearson's chi-squared test, Mann-Whitney U, and independent samples t-test, where appropriate. κ statistic was used to assess agreement between proportionality formulas. Kaplan-Meier survival curves were plotted for each group of patients. Patients were censured if mitral intervention or heart transplant/left ventricular assist device was performed. The log-rank test was used to assess for significant differences in time to endpoint between groups. Univariate and multivariate analysis with Cox regression were applied to assess the association between studied variables and all-cause mortality. Variables with a p<0.05 were included in the multivariable model. Net reclassification index (categorical NRI) was used to ascertain whether proportionality enhances the prognostic value of MR grade stratification according to ASE guidelines. All reported p-values are two-tailed, with a p=0.05 indicating statistical significance. Analysis was performed using IBM SPSS Statistics software, version 25 (2017).
ResultsStudy population and follow-upA total of 572 patients (mean age 69±12 years; 76% male) were included. The majority of patients were in class II (55.8%) or III (33.4%) of New York Heart Association (NYHA). There were 526 (92.0%) patients treated with beta blockers, 525 (91.8%) with angiotensin converting enzyme inhibitor/angiotensin II receptor blocker (ACEi/ARB), 235 (41.1%) with mineralocorticoid receptor antagonist, 170 (29.7%) had an implantable cardiac defibrillator (ICD), and 167 (29.2%) were under cardiac resynchronization therapy (CRT). Mean LVEF was 33±9%, with a median LVEDV of 174 mL (interquartile range (IQR): 136-220), and a median EROA of 14 mm2 (IQR: 8-22). According to ASE guidelines sMR was classified as mild (grade I) in 418 (73%) patients, moderate (grade II) in 109 (19%), moderate-to-severe (grade III) in 33 (6%) and severe (grade IV) in 12 (2%). Other clinical characteristics and echocardiographic parameters are further described in Table 1.
Clinical characteristics at baseline.
| Clinical characteristics | Total population (n=572) | Non-Events (n=318) | Events (n=254) | p-value |
|---|---|---|---|---|
| Age | 69±12 | 65±12 | 72±11 | <0.001 |
| Male | 434 (76) | 231 (73) | 203 (80) | 0.043 |
| Atrial fibrillation | 245 (43) | 113 (36) | 132 (52) | <0.001 |
| Hypertension | 414 (72) | 213 (67) | 201 (79) | 0.001 |
| Diabetes mellitus | 178 (31) | 95 (30) | 83 (33) | 0.472 |
| Creatinine (mg/dL) | 1.17 (0.92-1.82) | 1.07 (0.88–1.45) | 1.44 (1.02-2.59) | <0.001 |
| NYHA | <0.001 | |||
| I | 55 (9.6) | 45 (14) | 10 (4) | |
| II | 319 (55.8) | 188 (59) | 131 (52) | |
| III | 191(33.4) | 82 (26) | 109 (43) | |
| IV | 7 (1.2) | 3 (1) | 4 (2) | |
| Etiology | ||||
| Ischemic | 350 (61.2) | 182 (57) | 86 (66) | 0.030 |
| Non-ischemic | 222 (38.8) | 136 (61) | 86 (39) | 0.030 |
| Therapeutics | ||||
| ACEi/ARB | 525 (91.8) | 305 (96) | 220 (87) | <0.001 |
| Beta blockers | 526 (92.0) | 299 (94) | 227 (89) | 0.042 |
| MRA | 235 (41.1) | 134 (42) | 101 (40) | 0.566 |
| Diuretics | 368 (64) | 184 (58) | 184 (72) | <0.001 |
| ICD | 170 (29.7) | 113 (36) | 57 (22) | 0.001 |
| CRT-D/P | 167 (29.2) | 76 (24) | 91 (36) | 0.002 |
| Echocardiographic findings | ||||
| LVEDV (mL) | 169 [132-215] | 169 [131-216] | 178 [140-223] | 0.020 |
| LVEF (%) | 35 [28-40] | 36 [29-42] | 32 [24-38] | <0.001 |
| EROA (mm2) | 14 [8-22] | 12 [7-19] | 16 [10-24] | <0.001 |
| RVol (mL) | 23 [12-34] | 19 [11-30] | 27 [16-37] | <0.001 |
| RF (%) | 40 [22-60] | 34 [18-51] | 47 [31-70] | <0.001 |
| E | 0.84 [0.66-1.04] | 0.85 [0.69-1.00] | 0.94 [0.72-1.10] | <0.001 |
| E/e’ | 13 [10-17] | 12 [10-17] | 14 [11-18] | 0.007 |
| LAVI | 53 [41-70] | 50 [39-66] | 62 [51-76] | <0.001 |
| SPAP (mmHg) | 39 [33-49] | 38 [32-45] | 46 [37-57] | <0.001 |
| TAPSE (mm) | 19 [15-21] | 20 [17-22] | 17 [15-20] | <0.001 |
| TR≥moderate | 93 (16.3) | 34 (11) | 59 (23) | <0.001 |
| ASE sMR classification | <0.001 | |||
| Grade I | 418 (73) | 256 (80) | 162 (64) | |
| Grade II | 109 (19) | 43 (14) | 66 (26) | |
| Grade III | 33 (6) | 16 (5) | 17 (7) | |
| Grade IV | 12 (2) | 3 (1) | 9 (3) | |
ACEi: Angiotensin converting enzyme inhibitor; ARB: Angiotensin II receptor blocker; ASE: American Society of Echocardiography; CRT: Cardiac resynchronization therapy; EROA: Effective regurgitant orifice area; sMR: secondary mitral regurgitation; ICD: Implantable cardiac defibrillator; LVEF: Left ventricular ejection fraction; LVEDV: Left ventricular end-diastolic volume; MRA: Mineralocorticoid receptor antagonist; NYHA: New York Heart Association; RF: Regurgitant fraction; RVol: Regurgitant volume; SPAP: Systolic pulmonary artery pressure; TAPSE: Tricuspid annular plane systolic excursion;.
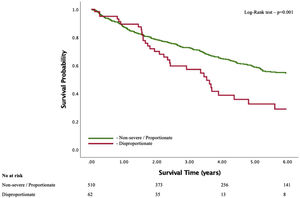
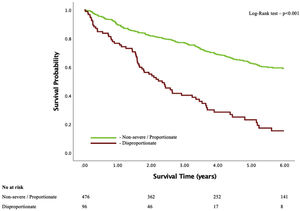
According to Grayburn's model, 62 patients (11%) were classified as disproportionate, and 510 (89%) as non-severe/proportionate. On the other hand, Lopes’ model categorized 96 (17%) patients as disproportionate and 476 (83%) as non-severe/proportionate. Amongst the 96 patients with disproportionate sMR Lopes’ formula, 46 (48%) were considered proportionate using Grayburn's formula; and of the 62 patients with disproportionate sMR according to Grayburn's, 12 (19%) were considered proportionate by Lopes’ (meaning only a moderate agreement between the two frameworks as depicted by Cohen's κ=0.58; p<0.001).
Different secondary mitral regurgitation proportionality frameworks with different prognostic valueDuring a mean follow-up of 4.1±2.7 years, there were 254 (44.4%) deaths. Kaplan-Meier survival curves for both models showed significant association between the presence of disproportionality and all-cause mortality (Figures 1 and 2). However, and after adjusting for several confounding variables associated with prognosis (namely, age, sex, creatinine, hypertension, atrial fibrillation, ischemic etiology, NYHA functional class, beta blockers, ACEi/ARB, diuretics, ICD, CRT, LVEDV, LVEF, E, E/e’, left atrial volume index, systolic pulmonary artery pressure, TAPSE and TR≥moderate), only Lopes’ formula maintained independent prognostic impact (adjusted HR 1.5 (95% CI 1.07-2.1), p=0.018; vs. Grayburn's formula: adjusted HR 1.0 (95% CI 0.67-1.5), p=0.998) – Table 2.
Hazard ratios (univariate and multivariate analysis) for the primary outcome.
| Hazard ratio (95% CI) | p | |
|---|---|---|
| Grayburn Model | ||
| Disproportionate sMR (univariate analysis) | 1.846 (1.289-2.643) | 0.001 |
| Disproportionate sMR (multivariate analysis*) | 0.999 (0.669-1.493) | 0.998 |
| Lopes Model | ||
| Disproportionate sMR (univariate analysis) | 3.174 (2.390-4.216) | <0.001 |
| Disproportionate sMR (multivariate analysis*) | 1.499 (1.072-2.097) | 0.018 |
CI: Confidence interval; sMR: secondary mitral regurgitation.
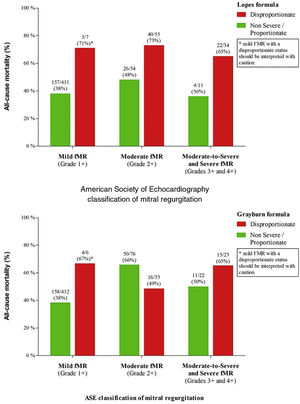
The reclassification of ASE SMR severity is presented in Figure 3 and Table 3. Only Lopes’ model was able to distinguish lower and higher risk subsets of patients according to the proportionality status when added to ASE SMR classification (NRI=0.129; p<0.001). Grayburn's model for disproportionality showed a non-significant NRI of 0.003 (p=0.455) when added to the ASE sMR classification and did not improve risk prediction.
Classifying sMR remains challenging, not only due to the morphology of the regurgitant orifice, but also owing to its dependence on loading conditions, its dynamic nature and its variation during the cardiac cycle. To the best of our knowledge, this is the first study that compared two different frameworks proposed to define sMR disproportionality. In this cohort, only Lopes’ model of disproportionate sMR was independently associated with all-cause mortality and showed improvement in risk stratification when added to the ASE classification.
Whether MR acts as a “bystander” or a “contributor” in HF patients with reduced LVEF has always been questioned. This debate has increased with the conflicting results of the two landmark trials of TEER in sMR. While MITRA-FR results suggest that sMR is just a “bystander”, COAPT findings seem to indicate that there is a subgroup of patients where sMR is a “contributor” to the disease which, when managed can lead to better outcomes.9,10 It became paramount to understand why both trials revealed conflicting results in a somewhat identical population of HF patients.3,4,9 Among the several explanations proposed by different investigators, the most striking one was the difference noted in mean EROA and LVEDV: MITRA-FR enrolled patients with smaller mean EROA and larger mean LVEDV when compared to COAPT.11 The concept of disproportionate sMR (which simultaneously accounts for the severity of MR and LVEDV) emerged as a potential identifier of patients where sMR is the primary driver (or “contributor”) of the progression of HF that might benefit the most from TEER.5–7
In this observational study, we sought to compare the prognostic impact of two published disproportionate sMR models and assess whether they could improve our current method of sMR classification. Even though various retrospective studies have been published showing the prognostic impact of Grayburn's model,6,12–14 our data suggest that the comparison of theorical EROA vs. measured EROA (Lopes’ formula model) has a greater association with all-cause mortality than the framework proposed by Grayburn. Two main reasons may justify these discrepancies: The cutoff of 0.14 proposed by Grayburn et al. only applies to patients with LVEF of 30%; Lopes’ model uses an individualized formula that is more versatile and can be applied to any patient irrespective of their LVEF (which is accounted for in the original formula).
Our study also shows that including proportionality status significantly improved the risk stratification of ASE sMR classification. While the presence of disproportionate sMR using Lopes’ formula showed a higher mortality risk for every ASE subgroup, this did not happen with Grayburn's model. This finding is particularly important for patients with at least moderate sMR, in whom the hemodynamic severity of the regurgitant lesion might not be accurately reflected by EROA and/or RVol alone. The inclusion of proportionality evaluation (which integrates LV size, LVEF and sMR grading) as an additional tool in a multiparametric assessment of sMR could be an important upgrade to identify patients at higher risk of events during follow-up. Nevertheless, it remains to be established (for example by a randomized clinical trial) whether patients with disproportionate sMR are indeed the subgroup that will benefit most from mitral intervention.
LimitationsOur study has some limitations that should be acknowledged. This was a retrospective, single center study, and our conclusions should be viewed as hypothesis generating due to the possible presence of unmeasured confounding factors and selection bias, and should be further validated in prospective studies. Also, the method used for evaluation of MR severity was two dimensional (2D) echocardiography, which has various limitations and imperfect concordance of severity among measured parameters.15,16 The regurgitant orifice in sMR is frequently semilunar or elliptical, thus affecting measurements and leading to possible underestimation of the EROA using the 2D PISA method. Furthermore, since volume status and blood pressure were not readily available on electronic records, they were not taken into account in the interpretation of sMR severity, which is notoriously dynamic. Moreover, caution should be taken when this concept is applied to patients with mild MR where measurement errors may be present.
ConclusionIn this study, the presence of disproportionate sMR was associated with lower survival during long term follow-up. Despite the existence of two formulas to define sMR proportionality status, only the Lopes et al. model maintained independent prognostic impact and managed to improve risk stratification for sMR severity according to the ASE criteria.
Author contributionConception and design (JP, PL, PF, FA, RR), data collection (JP, PL, PF, FA, CR, EH, MT, JA, PAG, MA, MJA, RR), data analysis (JP, PL, PF, FA, LM, SG, RR), drafting (JP, PL, PF, FA, RR), revising (JP, PL, PF, FA, CR, EH, LM, SG, MT, JA, PAG, MA, MJA, MM, RR). All authors read and approved the final manuscript.
Conflicts of interestThe authors have no conflicts of interest to declare.