Doxorubicin (DOX) is an anthracycline cytotoxic chemotherapeutic drug that is commonly used in cancer treatment. A major side effect limiting the clinical use of DOX is cardiotoxicity due to oxidative injury. Nigella sativa (NS) is an annual flowering plant with antioxidant properties. Its seeds contain several bioactive constituents such as saturated and unsaturated fatty acids, thymoquinone, dithymoquinone, thymohydroquinone, and thymol. In this study, we investigated the effect of NS extract on DOX-induced cardiotoxicity.
MethodsThe experimental study animals consisted of 28 male Sprague Dawley rats weighing between 300 and 400 g. Four study groups each of seven rats were defined: controls; NS extract; DOX; and DOX+NS. Control and DOX rats received standard food, while each rat in the NS and DOX+NS groups also received 100 mg/kg NS extract orally. At day 28 of follow-up, rats in the DOX groups were administered a single 10 mg/kg intraperitoneal dose of DOX, while rats in the control and NS groups received a single 10 mg/kg dose of physiological saline solution. All animals were monitored for 35 days. On day 35, the rats were decapitated and serum and cardiac tissue samples were obtained. Troponin and NT-proBNP levels were measured in blood sera, while malondialdehyde (MDA), nitric oxide, total antioxidant capacity (TAC), and total oxidative stress (TOS) levels were quantified in sera and tissue samples. Histological alterations that were assessed in cardiac tissue included myocyte disarray, small vessel disease, myocyte hypertrophy, and fibrosis.
ResultsThe DOX group had significantly higher NT-proBNP, TOS, and MDA, with greater histopathological derangement. TAC was significantly elevated in the DOX+NS group, which also exhibited significantly lower troponin, TOS, and MDA, as well as significantly higher TAC compared to the DOX group. Histopathological examination showed that the significant structural derangement observed in DOX rats was markedly and significantly reduced in DOX+NS rats.
ConclusionOur results suggest that NS extract may prevent DOX-induced cardiotoxicity and thus represents a promising cardioprotective agent.
A doxorrubicina (DX) é uma antraciclina citotóxica que é habitualmente utilizada no tratamento do cancro. Um efeito colateral major que limita a utilização clínica da DX é a cardiotoxicidade devida a lesão oxidativa. A Nigella sativa (NS) é uma planta de floração anual com propriedades antioxidantes. Contém diversos constituintes bioativos como os ácidos gordos saturados e insaturados, a timoquinona, a ditimoquinona, a timoidroquinona e o timol. Neste estudo, estudámos o efeito do extrato da NS na prevenção da cardiotoxicidade induzida pela doxorrubicina.
Material e métodosOs animais utilizados neste estudo experimental consistiram em 28 ratos Sprague Dawley do género masculino com peso entre 300 e 400 g. Foram definidos quatro grupos de estudo, com sete ratos em cada: controlos; extrato NS; doxorrubicina (DX); e DX+NS. Os ratos do grupo controlo e do grupo DX receberam alimentação padrão, enquanto cada rato dos grupos NS e DX+NS também recebeu 100 mg/kg de extrato de NS por via oral. No 28.° dia de seguimento, aos ratos dos grupos de doxorubicina foi administrada uma dose única de 10 mg/kg intraperitoneal de doxorubicina, enquanto os ratos dos grupos de controlo e de NS receberam 10 mg/kg de solução salina fisiológica de dose única. Todos os animais foram monitorados durante 35 dias. No 35.° dia, os ratos foram decapitados, tendo sido obtidas amostras de soro e de tecido cardíaco. Os níveis de troponina e de NT-pro BNP foram medidos no soro, enquanto os níveis de malondialdeído, de óxido nítrico, da capacidade antioxidante total e dos níveis totais de stress oxidativo foram quantificados em soros e amostras de tecido. As alterações histológicas que foram avaliadas no tecido cardíaco incluíram desalinhamento de miócitos, doença de pequenos vasos, hipertrofia miocitária e fibrose.
ResultadosO grupo DX apresentou NT-proBNP, stress oxidativo total (SOT) e malondialdeído (MAD) significativamente mais elevados, com maiores alterações histopatológicas. A capacidade antioxidante total (CAT) foi significativamente mais elevada no grupo DX+NX que também apresentou valores de troponina, SOT, e DAM significativamente mais baixos, assim como valores de CAT significativamente mais elevados em comparação com o grupo DX. Os exames histopatológicos mostraram que a disfunção estrutural significativa observada nos ratos DX foi marcadamente e significativamente menor nos ratos DX e NS.
ConclusãoOs nossos resultados sugerem que o extrato de Nigella sativa pode prevenir a cardiotoxicidade induzida pela doxorrubicina, representando um agente cardioprotetor promissor.
Cardiotoxicity, an important side effect of chemotherapeutic agents used for the treatment of cancer, is particularly important in the case of anthracyclines. Doxorubicin (DOX), an anti-tumor antibiotic of the anthracycline class, is frequently used in the management of carcinomas, sarcomas, and hematologic malignancies.1 Cardiotoxicity is a major factor limiting the clinical utility of DOX. Although the exact cause of DOX-induced cardiotoxicity remains elusive, many studies have implicated the role of oxidative stress, particularly due to the effects of free oxygen radicals as well as lipid peroxidation. One purported mechanism for cardiotoxicity involves failure of the antioxidant capacity of cardiac myocytes to counteract mitochondrial injury and lipid peroxidation caused by reactive oxygen species.2
Antioxidant treatments, including herbal antioxidants, have been used and tested in attempts to decrease the adverse consequences of oxidative stress. Nigella sativa (NS) is one such plant with antioxidant properties, the seeds of which are rich in saturated and unsaturated fatty acids as well as quinine-type compounds such as thymoquinone, thymohydroquinone, and dithymoquinone.3–6 Active components in the composition of NS seeds include amino acids, saturated and unsaturated fats (31-35.5%), tannins, minerals (phosphate, calcium and zinc), alkaloids, carbohydrates (33-34%), and vitamins (folic acid, thiamine, ascorbic acid and niacin).7 NS exerts its anti-inflammatory and antioxidant effects mainly through its quinine constituents.3–6 Specifically, the antioxidant effects of quinines (particularly thymoquinone) are based on the scavenging of superoxide radical ions and hydroxyl radicals as well as the prevention of lipid peroxidation.8,9
In this study, we investigated the cardioprotective effects of NS in DOX-induced cardiotoxicity in an experimental rat model.
MethodsThe experimental protocol used in the study was approved by the ethics committee of Firat University for animal experimentation. A total of 28 male Sprague Dawley rats aged 3-4 months and weighing between 300 and 400 g were used. Four experimental groups were defined with seven rats in each cage. Animals were fed with standard food and water ad libitum. The room temperature was maintained at 22±0.5°C. In order to prevent the possible effects of sex difference on the results and considering that male rats are more resistant, only male rats were used. Before the study, echocardiography was performed on the rats to be included in the study, which showed that they had normal cardiac function.
Experimental protocolThe seed of the NS plant was used in our study. 50 g of NS seed was kept in 400 ml hexane for 24 hours, then filtered and dried. The dried NS was extracted with methanol in a Soxhlet apparatus. Methanol in the extract was evaporated with a vacuum evaporator.10 In the chemical analysis of the extract used, thymoquinone (14.96%), thymol (0.06%), carvacrol (0.05%), p-cymene (0.86%), palmitic acid (12.08%), oleic acid (20.42%), linoleic acid (30.60%), linolenic acid (1.96%) and erucic acid (3.37%) were detected.
Rats in the control group (n=7) received standard nutrition and were followed for 35 days. On day 28, a single 10 mg/kg intraperitoneal dose of physiological saline was administered.
Rats in the DOX group (n=7) received standard nutrition and were followed for 35 days. On day 28, a single 10 mg/kg intraperitoneal dose of DOX was administered. The dose of DOX used was determined according to previous animal experimental models.11
Animals in the NS extract group (n=7) were given standard food together with a daily 100 mg/kg dose of NS extract diluted in water and administered through an orogastric tube for 35 days. On day 28, a single 10 mg/kg intraperitoneal dose of physiological saline was administered. In previous studies, an oral dose of 100 mg/kg of NS extract has been shown to prevent organ toxicity.12,13
Animals in the NS extract and DOX group (DOX+NS; n=7) were given standard food together with a daily 100 mg/kg dose of NS extract diluted in water and administered through an orogastric tube for 35 days. On day 28, a single 10 mg/kg intraperitoneal dose of DOX was administered.
On day 35, the rats were anesthetized with 0.5 mg/kg ketamine hydrochloride plus 0.1 mg/kg of xylazine hydrochloride. After intracardiac blood samples were taken, they were decapitated.
Preparation of specimensAfter the rats were decapitated, the cardiac tissue was removed, weighed, and fixed in 10% formaldehyde. Following fixation, 1-cm transverse sections were obtained from apex to base and embedded in paraffin blocks. Sections of 3-4 μm thickness were then prepared and examined after staining with hematoxylin-eosin and Masson trichrome.
Assessment of biochemical parametersDuring decapitation, 5 cc of blood samples were obtained in plain laboratory tubes. A 10-min period was allowed for coagulation, followed by centrifugation at 5000 rpm for 3 min. The separated serum was transferred to Eppendorf tubes and kept at -20°C until the time of analysis. Troponin and creatine in the serum sample were measured using an Olympus 2700 auto-analyzer (Olympus Co. Ltd., Tokyo, Japan). For N-terminal pro-B-type natriuretic peptide (NT-proBNP) measurements, an IMMULITE 2000 system was used (Siemens Healthcare Diagnostics, USA). For oxidative stress and antioxidant parameters, tissue samples were first weighed and then homogenized and oxidant parameters (total oxidative stress [TOS] and malondialdehyde [MDA]) and antioxidant parameters (total antioxidant capacity [TAC] and nitric oxide [NO]) were measured in serum and cardiac samples. MDA levels were determined using the method based on the reaction of MDA with thiobarbituric acid.14 TOS15 and TAC16 assays were based on the automated measurements developed by Erel. NO was measured with a commercial nitrate/nitrite colorimetric assay kit (catalog no. 780001; Cayman Chemical, Ann Arbor, MI, USA).
Histopathological assessmentsMyocyte disarray, myocyte hypertrophy, and small vessel disease were assessed in tissue cross-sections stained with hematoxylin and eosin. Fibrosis was assessed using Masson trichrome staining in samples. These parameters were graded as follows: 0, absent; 1, mild; 2, moderate; and 3, severe. The histopathological assessments were scored blindly.
Statistical analysisIBM SPSS for Windows v. 22.0 was used for data analysis. All data were expressed as mean ± standard deviation. The four experimental groups were compared and since the variances were homogeneously distributed, multiple groups were compared using one-way analysis of variance (ANOVA) with post-hoc Tukey correction. A p-value<0.05 was considered statistically significant.
ResultsAll experimental animals survived until the completion of the study.
Serum biochemistry parametersRats in the DOX group had significantly elevated troponin compared to all other groups. Also, NT-proBNP was significantly higher among DOX rats compared to the control and NS groups. Troponin was significantly higher in DOX+NS rats than in controls and NS rats, while it was significantly lower than in DOX rats. NT-proBNP in DOX+NS rats was lower than in the DOX group, although the difference did not reach significance. Results of serum biochemistry assessments are shown in Table 1.
Serum biochemistry parameters in experimental rat groups.
| Controls (n=7) | NS (n=7) | DOX (n=7) | DOX+NS (n=7) | |
|---|---|---|---|---|
| Troponin, ng/ml | 0.12±0.01 | 0.33±0.17 | 3.47±0.49a | 1.64±0.26b |
| NT-proBNP, pg/dl | 26.71±2.78 | 25.28±3.01 | 49.42±6.26c | 34.71±3.38 |
| Creatine, mg/dl | 0.47±0.01 | 0.51±0.01 | 0.44±0.02 | 0.41±0.02 |
DOX: doxorubicin; NS: Nigella sativa extract; NT-proBNP: N-terminal pro-B-type natriuretic peptide.
TOS and MDA levels in DOX rats were significantly higher than in all other groups. TOS in DOX+NS group was significantly higher than that in the NS group, while it was significantly lower than in the DOX group. TAC in the DOX+NS group was significantly higher than in all other study groups. NO in DOX+NS rats was significantly higher compared to control and NS groups. Serum oxidant and antioxidant status in the study groups is shown in Table 2.
Oxidant and antioxidant parameters in experimental rat groups.
| Controls (n=7) | NS (n=7) | DOX (n=7) | DOX+NS (n=7) | |
|---|---|---|---|---|
| TOS, μmol/l | 52.37±8.68 | 45.75±7.03 | 332.27±35.99a | 176.06±20.02b |
| TAC, mmol/l | 0.85±0.14 | 2.35±0.43 | 3.02±0.44 | 7.87±1.04c |
| NO, μM | 4.71±1.06 | 4.57±0.42 | 56.86±9.23 | 75.14±6.58d |
| MDA, mmol/ml | 34.14±3.75 | 29.71±4.41 | 134.57±25.81e | 72.00±6.97 |
DOX: doxorubicin; MDA: malondialdehyde; NO: nitric oxide; NS: Nigella sativa extract; TAC: total antioxidant capacity; TOS: total oxidative stress.
TOS was significantly higher in the DOX group compared to all other groups. Also, MDA in the DOX group was significantly elevated versus controls. TAC in DOX+NS rats was significantly higher in comparison with other study groups. NO in the DOX+NS group was significantly higher than in control and NS groups. Although NO in the DOX+NS group was higher than in the DOX group, the difference did not reach statistical significance. Table 3 shows tissue oxidant and antioxidant status.
Tissue oxidant and antioxidant status in experimental rat groups.
| Controls (n=7) | NS (n=7) | DOX (n=7) | DOX+NS (n=7) | |
|---|---|---|---|---|
| TOS, μmol/l | 214.60±41.13 | 215.46±16.82 | 425.36±42.65a | 280.77±40.56 |
| TAC, mmol/l | 4.53±1.22 | 8.09±1.24 | 18.57±3.56c | 31.03±4.67b |
| NO, μM | 36.57±4.70 | 48.86±6.45 | 85.57±8.46c | 130.29±21.26d |
| MDA, mmol/ml | 101.43±5.89 | 95.71±5.12 | 154.71±21.62c | 135.29±13.45 |
DOX: doxorubicin; MDA: malondialdehyde; NO: nitric oxide; NS: Nigella sativa extract; TAC: total antioxidant capacity; TOS: total oxidative stress.
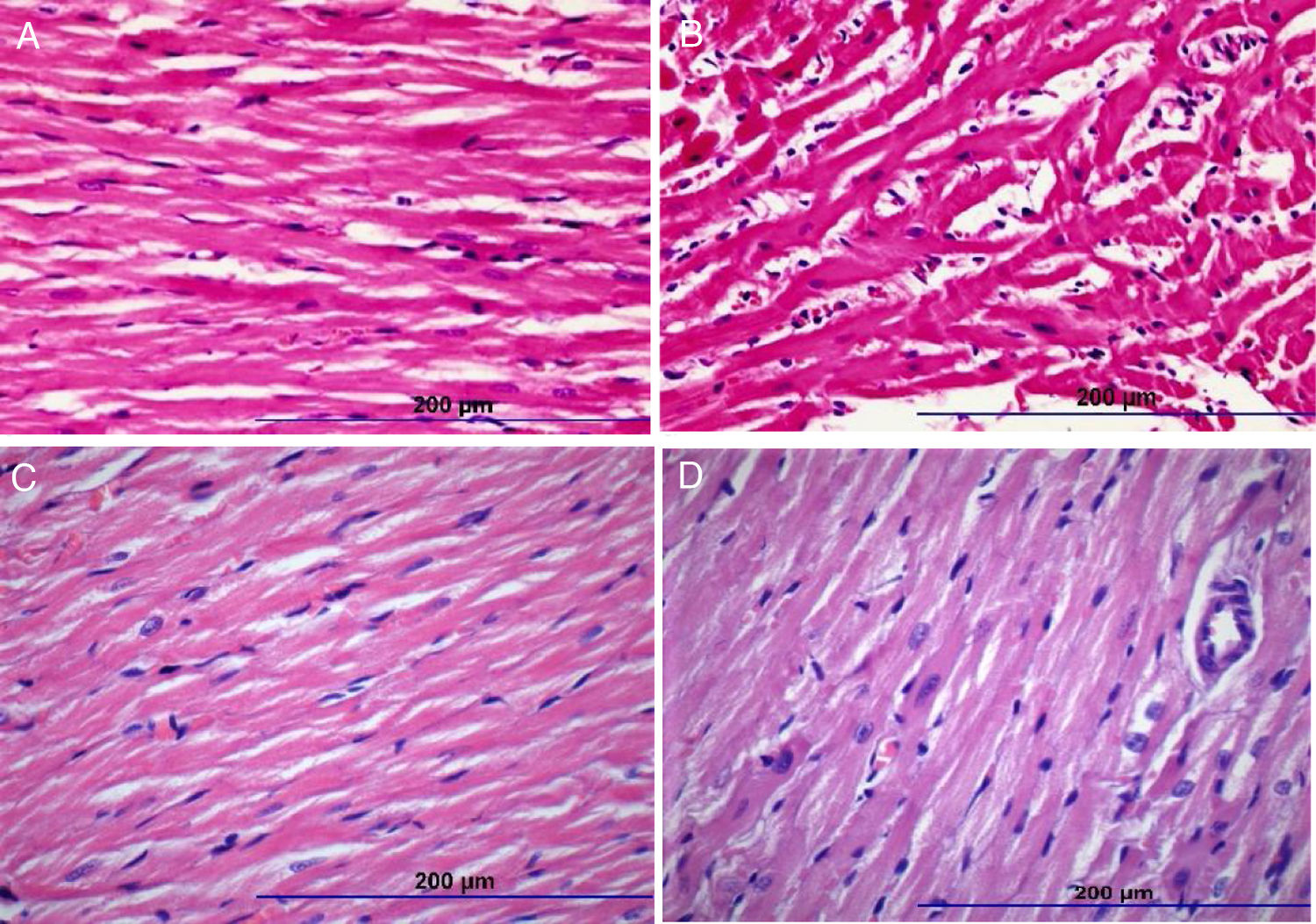
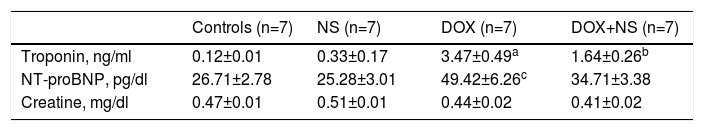
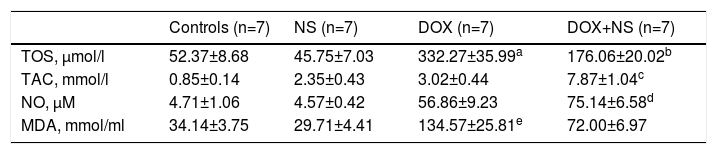
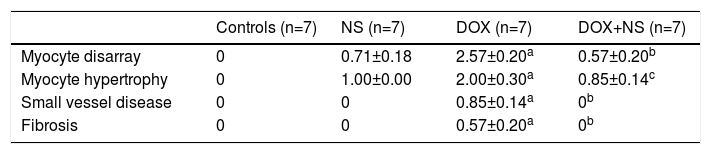
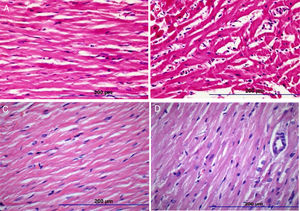
There were significant alterations in all histopathological parameters (myocyte disarray, myocyte hypertrophy, small vessel disease, and fibrosis) in the DOX group compared to controls. On the other hand, myocyte disarray, myocyte hypertrophy, small vessel disease, and fibrosis were significantly lower in the DOX+NS group compared to the DOX group. Histopathological findings are summarized in Table 4 and Figure 1.
Histopathologic images of study groups (hematoxylin and eosin, ×200). (A) Normal myocyte alignment in the control group (n=7); (B) severe myocyte disarray and hypertrophy in the DOX group (n=7); (C) normal myocyte alignment in the NS group (n=7); (D) significantly less myocyte disarray and hypertrophy in DOX+NS rats vs. DOX rats (n=7).
DOX is a chemotherapeutic agent belonging to the class of anthracycline cytotoxic antibiotics,17 and is commonly used for the treatment of carcinomas, sarcomas, and hematologic malignancies. As well as its therapeutic action on tumor cells, DOX can also cause serious side effects via toxicity in healthy tissues.18 One such adverse effect is cardiotoxicity. Although numerous mechanisms for the pathogenesis of cardiotoxicity have been put forward, most studies suggest a key role for lipid peroxidation, increased reactive oxygen species, induction of oxidative stress, and reduced antioxidant levels.19 The mitochondrial membrane is thought to be the main target of anthracycline-related cardiotoxicity. Heart tissue produces most of its energy in the mitochondria through beta-oxidation of fats. Studies have shown that DOX tends to accumulate in mitochondria.20 It has been reported that as a result of DOX accumulation and toxicity in the tissue, there is a decrease in ATP production together with apoptosis as a result of morphological and functional disorders in mitochondria.20,21 High NO concentrations resulting from activation of inducible NO synthase may cause DOX cardiotoxicity.20–22 In our study, the significantly higher NO levels in the DOX group with cardiotoxicity compared to the control group are supported by previous studies. On the other hand, enhanced antioxidant parameters can help prevent toxicity resulting from oxidative stress. In this regard, it should be noted that several antioxidants of plant origin have been shown to prevent cytotoxicity due to oxidative stress.23,24
NS is one such plant with antioxidant properties, its constituents including saturated and unsaturated fatty acids, thymoquinone, dithymoquinone, thymohydroquinone, and thymol.25,26 The antioxidant effects of NS and these ingredients have been documented in several studies.27 Quinine compounds are particularly important for the anti-inflammatory and antioxidant effects of NS.3–6 In a study by Ojha et al., thymoquinone, a constituent of NS, was shown to confer cardioprotective effects against isoproterenol-induced myocardial injury via reduced oxidative stress and inflammation.28 Also, Nagi et al. observed that administration of thymoquinone suppresses elevation of CK-MB due to cardiotoxicity.29 Similarly, in our study, the addition of NS to DOX resulted in a significant reduction in levels of the cardiac injury marker troponin.
In our study, DOX was associated with cardiac injury, as reflected by the significant increase in troponin and NT-proBNP levels. However, rats in the DOX+NS group had considerably lower troponin levels than DOX rats, while a near-significant decline was also observed in NT-proBNP. In terms of histological parameters, myocyte disarray, myocyte hypertrophy, small vessel disease and fibrosis scores were significantly higher in DOX rats than in the other groups. However, the addition of NS to DOX resulted in a significant alleviation of these histological changes compared to the DOX group. In parallel with these observations, rats in the DOX group presented significant increases in serum TOS and MDA oxidant levels in comparison with other study groups. By contrast, serum TAC (reflecting antioxidant status) was significantly higher, and TOS and MDA (both reflecting oxidant status) were significantly lower in the DOX+NS group. Similarly, when tissue oxidant and antioxidant parameters are considered, rats in the DOX group had significantly elevated TOS compared to all other groups, and TAC was significantly higher in DOX+NS rats than in DOX rats. These results show that DOX causes cardiac injury in conjunction with increases in oxidative parameters, while the addition of NS to DOX resulted in a decrease in oxidative parameters, improvement of antioxidant status, and significantly less pronounced histopathological cardiac injury.
In our study, significantly higher values (p<0.05) of myocyte disarray and myocyte hypertrophy and also a slight increase in troponin levels were observed in the NS group compared to the control group. However, the high NO levels seen in the NS and DOX+NS groups indicate that NS may also have proinflammatory properties. In a study by Islam et al., NS reduced neutrophil and splenocyte counts and increased peripheral lymphocyte and monocyte levels.30 Cases of allergic contact dermatitis with maculopapular eczema symptoms have been reported following topical use of pure NS oil.31 However, it has been reported that the toxic effect of NS is low. In vivo studies have shown that high doses of thymoquinone cause liver damage.31,32 The results of our study and the findings of similar studies confirm that very high doses of NS have proinflammatory and toxic effects.
ConclusionNS extract prevents cardiac injury through alleviation of DOX-induced cardiotoxicity and therefore appears to be a suitable agent for cardioprotection. Although a beneficial effect on cardiotoxicity is observed, the proinflammatory properties of the extract should be taken into account and lower concentrations should be considered.
Conflicts of interestThe authors have no conflicts of interest to declare.