To present the Portuguese results of a multi-country cross-sectional survey aiming to estimate productivity loss in the first year after an acute coronary syndrome (ACS) or stroke.
MethodsPatients previously hospitalized for ACS or stroke were enrolled during a routine cardiology/neurology visit 3-12 months after the index event and ≥4 weeks after returning to work. Productivity loss for the patient and the caregiver in the previous four weeks were reported by the patient using the validated iMTA Productivity Cost Questionnaire (iPCQ). Hours lost were converted into eight-hour work days and prorated to one year, combined with initial hospitalization and sick leave, and valued according to Portuguese labor costs.
ResultsThe analysis included 39 employed patients with ACS (mean age 51 years, 80% men, 95% with myocardial infarction, mean left ventricular ejection fraction 55%) and 31 with stroke (mean age 50 years, 80% men, all ischemic, 77% with modified Rankin Scale 0-1); 41% of ACS and 10% of stroke patients had a history of cardiovascular disease. Mean (SD) productivity loss for patients and caregivers was 47 (62) work days for ACS and 76 (101) work days for stroke. ACS patients lost 37 (39) and caregivers lost 10 (42) work days. Stroke patients and caregivers lost 65 (78) and 12 (38) work days, respectively. Total mean indirect cost per case was €5403 (€7095) and €8726 (€11558) for employed patients with ACS and stroke, respectively.
ConclusionsThe annual proportions of productive time lost by employed patients due to ACS and stroke in Portugal were 17% and 27%, respectively. Caregivers of these patients lost about 5% of their annual productive time.
Apresentar os resultados portugueses de um estudo de corte transversal desenvolvido em vários países que estima a perda de produtividade no primeiro ano após eventos de síndrome coronária aguda (SCA) ou de acidente vascular cerebral (AVC).
MétodoDoentes previamente hospitalizados por SCA ou AVC foram recrutados em consultas de cardiologia e neurologia 3-12 meses após índice de hospitalização por evento e quatro semanas após regresso ao trabalho. Perda de produtividade de doentes e cuidadores foi reportada pelos doentes nas quatro semanas anteriores utilizando o validado iMTA Productivity Cost Questionnaire (iPCQ). Horas perdidas foram convertidas em dias de trabalho com duração de oito horas e dimensionados a um ano, combinadas com hospitalização inicial e baixa médica e valorados segundo o custo laboral em Portugal.
ResultadosA análise incluiu doentes empregados, dos quais 39 sofreram SCA (51 anos, 80% homens, 95% enfarte do miocárdio, 55% fração de ejeção ventricular esquerda) e 31 sofreram AVC (50 anos, 80% homens, 100% doenças isquémicas do coração, 77% escala 0-1 modificada de Rankin); 41% dos doentes com SCA e 10% dos doentes com AVC tinham história de doenças cardiovasculares. A produtividade perdida média (DP) para doentes e cuidadores, em dias de trabalho, foi 47 (62) para SCA e 76 (101) para AVC. Doentes e cuidadores de SCA perderam 37 (39) e 10 (42) dias de trabalho, respetivamente. Doentes e cuidadores de AVC perderam 65 (78) e 12 (38) dias de trabalho, respetivamente. O custo total indireto foi €5,403 (€7,095) e €8,726 (€11,558) por doente empregado com SCA e AVC, respetivamente.
ConclusãoEm Portugal, as proporções anuais de tempo produtivo perdido para doentes empregados após SCA e AVC foram, respetivamente, 17% e 27%. Cuidadores destes doentes perderam cerca de 5% do seu tempo produtivo anual.
Cardiovascular disease (CVD) is responsible for about one third of global mortality every year, with 18 million CVD deaths in 2015 worldwide, of which 15 million deaths were due to ischemic heart disease and stroke.1 CVD was responsible for 29% of the 107000 deaths in Portugal in 2016.2 Although mortality from CVD in Portugal has decreased by more than 40% over the last 10 years,3 the number of patients discharged from the hospital having survived an event is not decreasing and therefore there are more patients alive with a history of the disease.3,4 Thus, the burden of CVD morbidity is expected to grow further, weighing heavily not only on healthcare systems and patients’ quality of life but also on work productivity, as well as increasing the need for informal care of people with CVD.
To our knowledge, existing information on productivity losses after CVD in Portugal is very limited.5 In a multi-country study, the direct and indirect costs of cardiovascular disease in Portugal for 2003 were estimated at around €1762 million, of which €969 million was healthcare costs.6 A more recent study conducted in the European Union in 2017 estimated the overall cost of CVD in Portugal at €2.8 billion a year, with direct healthcare costs accounting for 42% of total costs, work productivity losses for 27% and informal care for the remaining 31%.7
The aim of the current study was to estimate productivity loss and indirect costs for patients and caregivers in the first year after an acute coronary syndrome (ACS) or stroke in Portugal.
MethodsStudy design and populationPortugal was one of the seven European countries (the others were Belgium, France, Poland, Spain, Switzerland, and the UK) that participated in a multi-country cross-sectional study of patient and caregiver productivity loss and indirect costs following ACS or stroke in Europe. The results of this multi-country study are published elsewhere.8 Patients were recruited during a routine consultation at a cardiologist's or neurologist's clinic between September 2016 and November 2017. Inclusion criteria were hospitalization for ACS (myocardial infarction [MI] or unstable angina) or stroke 3-12 months prior to the recruitment visit, having a paid job, and receiving lipid-modifying therapy at the time of the hospitalization for the cardiovascular event (CVE). To assess absenteeism and presenteeism at work, patients had to be back to work for at least four weeks prior to recruitment. Patients who participated in a clinical trial at the time of recruitment or were treated with evolocumab were excluded from the study.
Patients who were able to return to work after an event were recruited in four clinical centers located in different parts of Portugal. Patients who were unemployed at the time of their event or those who did not return to work were not included in the analyses. The objective was to recruit 33 ACS and 33 stroke patients, to achieve a 34% precision in the cost estimate, assuming that sample mean and standard deviation (SD) are equal.
The study received approval from the appropriate hospital ethics committees and the Portuguese Data Protection Authority (CNPD). All participants provided written informed consent.
Data collectionPatient absenteeism, presenteeism and caregiver time were assessed using the IMTA Productivity Cost Questionnaire (iPCQ), a validated questionnaire developed by the Institute for Medical Technology Assessment, Erasmus University Rotterdam, The Netherlands, to collect productivity loss data.9 This questionnaire has a four-week recall period to overcome recall bias and robust costing methodology, and can be completed in about 10minutes, minimizing the additional burden to patients. Two additional questions were added to the iPCQ to assess absenteeism due to the index CVE hospitalization and sick leave immediately after discharge. The questions were reviewed by the iMTA team that developed the original questionnaire in order to maintain the integrity of the tool. The modified questionnaire was translated into Portuguese using forward-backward translation.
Demographic and clinical characteristics were abstracted by the recruiting physician from patients’ medical records and provided via an electronic case report form (eCRF). This eCRF also included data that enabled the length of the index hospitalization to be cross-checked against the data reported by the patient. Conflicting or implausible responses were reconciled directly with the recruiting centers.
AnalysisTo estimate annual patient productivity loss, the reported time loss for four weeks was prorated to the rest of the year and combined with the productive time lost due to the index hospitalization and sick leave. Similarly, annual caregiver loss was prorated from the reported loss for the four weeks preceding recruitment. Lost productive time during the year was calculated based on patient-reported working hours per day and considering Portuguese annual leave and bank holidays (a mean of 36 days in total). All calculations were performed at the patient level and summarized by ACS and stroke.
Productivity loss was valued using the human capital approach for the patient (i.e. assuming that each worker is irreplaceable for society so that all productive time lost by the worker is lost to society) and the opportunity cost for the caregiver (i.e. forgone production).4,10 Each hour lost was multiplied by the Portuguese labor cost taken from Eurostat. To obtain 2018 values, labor costs from 2012 to 2017 were projected using the geometric average (€14.27).11
The friction cost method (i.e. assuming that workers are replaceable and it takes a certain ‘friction’ period to find and train a replacement; after this friction period productivity losses are no longer incurred by society) was used in a sensitivity analysis with a friction period of three months.12
Descriptive statistics were expressed as frequency and percentage for categorical variables and mean (standard deviation) unless otherwise specified and ranges for continuous variables. Missing clinical values were not replaced; missing numbers of days lost were assumed to equal zero.
All analyses were performed using SAS® Statistical software (SAS Institute, Cary, NC) version 9.4.
ResultsPopulation characteristicsFour centers participated in the study: one in Faro, two in the Greater Lisbon area, and the fourth in Braga. A total of 39 patients with ACS and 31 patients with stroke were included across the four centers. The baseline characteristics of the study population are shown in Table 1.
Baseline demographic and clinical characteristics of the study population.
| ACS (n=39) | Stroke (n=31) | |
|---|---|---|
| Gender, male, n (%) | 31 (79.5%) | 25 (80.7%) |
| Age, years, mean (SD) | 51.3 (7.5) | 49.9 (8.5) |
| Urban residence, n (%) | 33 (84.6%) | 24 (77.4%) |
| Education level and employment, n (%) | ||
| Below secondary | 18 (46.2%) | 9 (29.0%) |
| Secondary | 17 (43.6%) | 15 (48.4%) |
| Higher education | 4 (10.3%) | 4 (12.9%) |
| White collar (office employee) | 22 (56.4%) | 16 (51.6%) |
| Blue collar (manual worker) | 17 (43.6%) | 15 (48.4%) |
| Type of ACS, n (%) | ||
| Myocardial infarction | 37 (94.9%) | NA |
| Unstable angina | 2 (5.1%) | NA |
| LVEF, %, mean (SD) | 54.5 (11.4) | NA |
| Revascularization, n (%) | 35 (89.7%) | NA |
| CABG | 1 (2.6%) | NA |
| PCI | 34 (87.2%) | NA |
| Type of stroke | ||
| Ischemic | NA | 31 (100.0%) |
| mRS at discharge, n (%) | ||
| 0 | NA | 7 (22.6%) |
| 1 | NA | 17 (54.8%) |
| 2 | NA | 5 (16.1%) |
| 3+ | NA | 2 (6.5%) |
| CV risk factors, n (%) | ||
| BMI, kg/m2, mean (SD) | 26.5 (4.2) | 26.8 (3.9) |
| Current smoker, n (%) | 21 (53.9%) | 9 (29.0%) |
| Type 2 diabetes, n (%) | 5 (12.8%) | 7 (22.6%) |
| Dyslipidemia, n (%) | 39 (100.0%) | 31 (100.0%) |
| LDL-C ≥70 mg/dl (1.8 mmol/l) | 36 (92.3%) | 27 (87.1%) |
| LDL-C ≥100 mg/dl (2.5 mmol/l) | 28 (71.8%) | 26 (83.9%) |
| Receiving high-intensity statin therapya | 30 (76.9%) | 17 (54.8%) |
| Hypertension, n (%) | 34 (87.2%) | 22 (71.0%) |
| SBP ≥140 mmHg | 8 (20.5%) | 3 (9.7%) |
| Previous CVE or established CV disease, n (%)b | 16 (41.0%) | 3 (9.7%) |
| CCI score, mean (SD) | 1.4 (1.1) | 1.5 (1.2) |
| CCI score ≥2, n (%) | 10 (25.6%) | 8 (25.8%) |
| Time since CVE, months, mean (SD) | 7.4 (2.5) | 6.5 (3.0) |
| Index CVE hospitalization, days, mean (SD) | 4.6 (4.2) | 8.6 (7.7) |
| CVE post-index hospitalization, n (%) | 3 (7.7%) | 0 (0.0%) |
Previous CVE or established CV disease: patients who had experienced myocardial infarction, unstable angina, hemorrhagic or ischemic stroke, transient ischemic attack, stable angina, carotid stenosis, congestive heart failure, peripheral vascular disease or cardiac ischemia before the index hospitalization.
ACS: acute coronary syndrome; BMI: body mass index; CABG: coronary artery bypass grafting; CCI: Carlson comorbidity index; CV: cardiovascular; CVE: cardiovascular event; LDL-C: low-density lipoprotein cholesterol; LVEF: left ventricular ejection fraction; m Rs: modified Rankin scale; NA: not applicable; PCI: percutaneous coronary intervention; SBP: systolic blood pressure; SD: standard deviation.
The mean age of ACS patients was 51 (7.5) years, 80% were men, 85% lived in urban areas and 56% were white-collar workers. Almost 95% of patients with ACS had an MI. However, the left ventricular election fraction of the MI patients was well preserved (mean 55%). All ACS patients had dyslipidemia, 54% were current smokers and 13% had type 2 diabetes. More than 40% of ACS patients had a history of a prior CV event. Patients were recruited on average seven months after their CVE. The ACS patients worked on average 37.1 (9.6) hours over 5.0 (0.9) days per week; mean duration of a work day was 7.5 (2.1) hours.
The mean age of stroke patients was 50 (8.5) years and 81% were men, 77% lived in urban areas and 52% were white-collar workers. All these patients suffered an ischemic stroke. In most cases the stroke had relatively little impact on mobility (77.4% of patients had modified Ranking scale [mRS] score ≤1). Seventy-four per cent of patients were hypertensive, 23% had type 2 diabetes and 9% had a history of prior CVEs. Stroke patients were recruited on average seven months after their CVE. These patients worked on average 37.8 (13.5) hours over 5.4 (0.7) days per week; mean duration of a work day was 7.0 (2.3) hours.
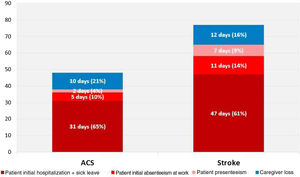
Productive time lostThe mean productivity loss by patients and caregivers during the first year after an ACS was 47.3 (62.2) work days; after a stroke patients and caregivers lost a mean of 76.4 (101.2) work days (Figure 1).
ACS patients lost a mean of 37.3 (38.5) work days due to absenteeism and presenteeism; caregivers lost an additional 10 (42.5) days helping an ACS patient. Stroke patients lost a mean of 64.7 (78.2) work days due to absenteeism and presenteeism and an additional 11.7 (37.9) work days were lost by caregivers helping a stroke patient.
On average, 82% of the time lost by ACS patients was due to absenteeism caused by the index hospitalization and initial sick leave. In stroke patients the index hospitalization and initial sick leave accounted for 73% of lost productive time. Presenteeism accounted for 4% (1.6 work days) in ACS patients and 11% (7.1 work days) in stroke patients (Figure 1).
Indirect costsIn the first year after the index CVE, the mean total indirect cost associated with patients’ and caregivers’ productivity loss was €5403 (€7095) for ACS and €8726 (€11558) for stroke (Table 2). Of those, €4259 (€4394) and €7386 (€8927), respectively, were lost by ACS and stroke patients. Caregivers’ indirect costs were €1144 (€4847) for ACS and €1340 (€4322) for stroke, respectively. Although the absolute numbers were slightly lower, the patterns of lost productivity remained unchanged when the friction cost method was used (Supplementary Table S1).
Annual costs by type of cardiovascular event (human capital method) in euros, mean (SD).
| ACS (n=39) | Stroke (n=31) | |
|---|---|---|
| Patient absenteeism | 4080 (4238) | 6582 (7877) |
| Initial hospitalization + sick leave | 3477 (3346) | 5363 (6237) |
| Absenteeism from work | 603 (2664) | 1219 (4832) |
| Patient presenteeism | 179 (591) | 805 (2331) |
| Caregiver loss | 1144 (4847) | 1340 (4322) |
| Total indirect costs | 5403 (7095) | 8726 (11558) |
ACS: acute coronary syndrome; SD: standard deviation.
In our study we observed substantial productivity losses and indirect costs in the first year following a CVE in Portugal, with 47 work days lost by patients and caregivers after an ACS and 76 work days after a stroke, and mean total indirect costs of €5403 and €8726, respectively.
About 80% of productive time in ACS patients was lost due to the patient's absence immediately following the CVE. This is much more than the average of 60% that was observed in the other six European countries that participated in this study.4 Once Portuguese patients returned to work they continued to lose productive time due to absenteeism (five work days for ACS and 11 work days for stroke patients) and presenteeism (two work days for ACS and seven work days for stroke patients). The loss of productive time observed in Portuguese patients was considerably lower than the average time lost in the other six countries in the project. After returning to work Portuguese ACS patients lost on average seven work days during the first year after the event, while stroke patients lost 18 work days. The corresponding losses in the other six European countries were 24 work days for ACS and 22 work days for stroke patients. A possible explanation for the lower productivity loss compared to France, Poland, Spain, Switzerland and the UK could be the proportion of blue-collar workers in the Portuguese sample (45% in Portugal vs. 26% in the other six countries). Blue-collar workers may be more concerned about losing their job for being on sick leave for too long or being unproductive. However, it should be noted that unemployment in Portugal, although higher than in Switzerland, UK, Poland and Belgium, is much lower than in France and Spain, where such low productivity losses were not seen after returning to work.13 ACS patients in Portugal tended to lose less productive time due to the initial hospitalization and sick leave compared to the other participating countries (31 vs. 37 days). Portuguese stroke patients, on the other hand, lost more time than did stoke patients in the other six countries that participated in the project (47 days vs. 31 days).
The considerable presenteeism that we observed in stroke patients could be explained by cognitive problems and fatigue after the event that are not considered in the mRS score. Although the Portuguese stroke population had the same proportion of stroke patients with mRS ≤ 1 as did the other countries in the project (77%), the distribution of patients between mRS = 0 and mRS = 1 was different. In the Portuguese population 55% had mRS=1 whereas in the overall study this population comprised a mean of 38%. This could explain the longer initial hospitalization and sick leave and longer period of absenteeism in Portuguese patients.
According to the 2016 literature review by Gordois et al., very little information is available in Portugal on indirect costs in general and on cardiovascular disease in particular.5 Gouveia Pinto reported that the estimated cost of MI for the first 12 weeks was €5450, which is very close to what we observed in ACS patients (€5403).14 For stroke our estimate of indirect costs seems to be double the direct cost as reported by Miguel et al. (€8726 vs. €4136).15
An important strength of our study is that, unlike most indirect costs studies, which focus on retrospective investigation of databases or administrative records, we collected information directly from CVE patients to capture information on absenteeism after return to work, presenteeism, and caregiver help. We used the most recent validated tool (iPCQ) specifically designed for assessing productivity losses over the previous four weeks – the longest period that can be assessed without recall bias having a significant effect.9
We recruited ACS and stroke patients who were still of working age, were employed prior to their event and were able to return to work afterward. This population is expected to be healthier and younger than the overall population of ACS and stroke patients. Therefore, our study provides a conservative estimate of productivity losses in ACS and stroke patients; the burden of productivity loss and indirect costs in the overall population of ACS and stroke patients is likely higher, as patients with more severe ACS and stroke may take more than 12 months to return to work (for example because they require longer rehabilitation) or in fact never return to work due to acquired disability.
The main study limitations are typical of all cross-sectional studies with patient-reported data. Our patients were recruited from four different centers. Although every attempt was made to select centers in different regions across the country, most of the centers served urban populations, leading to the possibility of rural populations being under-represented. These factors may limit the generalizability of our results to a broader ACS and stroke population in Portugal.
Prorating was based on individual absenteeism, presenteeism and caregiver loss patterns over the previous four weeks and assumed to remain constant between return to work and the end of the year. We analyzed the distribution of patients who were recruited within six months of the event and those who were recruited 6-12 months after the event, and found no difference in productivity loss patterns depending on the time elapsed since the event.
By focusing on employed patients only, we did not take into account caregiver time in patients who were unemployed or did not return to work. Since the overall population of MI and stroke patients does include these patients and their caregivers, the estimates for overall caregiver productivity losses are likely to be higher.
Concerning wages, salary-related differences between genders and age groups could have been considered to refine this value. However, given that this analysis was part of a multi-country project we opted to use the same source (Eurostat) for all the different countries, with the associated limitations.
For this analysis, a validated questionnaire (iPCQ) was used and instructions for users on how to translate the answers into costs were followed. For caregivers it is not known whether they were employed, and if so, whether they performed their caregiving tasks during working time.
ConclusionOur findings suggest that ACS and stroke were associated with substantial productivity loss and indirect costs in Portugal. ACS patients lost a mean of 17% of their work days during the first year after the event and stroke patients lost 27% of work days. In addition, caregivers lost 4% and 5% of their annual productive time helping ACS and stroke patients, respectively. In Portugal, productivity loss in terms of work days lost associated with ACS was lower than in the other six European countries that participated in the study. Productivity loss in Portuguese stroke patients was similar to that observed in the other participating countries.
The associated indirect costs, €5403 for ACS and €8726 for stroke, are comparable to the direct medical costs of these conditions as reported in earlier studies, and therefore potentially double the total cost of ACS and stroke from a societal perspective. Similar relationships between direct and indirect costs of ACS and stroke were found in all seven European countries that participated in the study.
Data sharing statementQualified researchers may request data from Amgen clinical studies. Complete details are available at http://www.amgen.com/datasharing.
Authors’ contributionsNM, HP, SR, AGF, ADA: contributed to patient recruitment, interpreted the results, critically reviewed the manuscript.
ES, LG, MR, RM: developed the study concept and design, coordinated the centers and patient recruitment, supervised the analyses, critically reviewed the manuscript.
LG: wrote the study protocol, programmed the analyses.
MR: drafted the manuscript.
FundingThis study was funded by Amgen (Europe) GmbH.
Conflicts of interestES is a full-time employee at Amgen (Europe) GmbH and owns Amgen stock options. RM is a full-time employee at Amgen Biofarmacêutica Lda (Portugal). IQVIA, employer of LG and MR, received consulting fees from Amgen (Europe) GmbH.
The authors would like to thank to all study investigators, study nurses and patients who participated in the study: Dr Ana Marques (Hospital Garcia de Orta, Lisbon), Dr Pedro Nascimento Alves (Hospital de Santa Maria, Lisbon), Dr João Diogo Pinto (Hospital De Braga), Dr Dina Bento (Hospital De Faro); Tim Kanters at iMTA for his methodological input on the study design and analyses; and iMTA in general for granting permission to use and translate the iPCQ questionnaire for our study. The authors would also like to thank David Gomez and Mark Lamotte for their ongoing support.