Levels of physical activity in modern urbanized society are clearly insufficient to maintain good health, and to prevent cardiovascular and other disease. Aerobic exercise is almost completely free of secondary effects, and is a useful adjunctive therapy in treating hypertension. There are several possible mechanisms to account for the beneficial effects of exercise in reducing blood pressure, the resulting physiological effects usually being classified as acute, post-exercise or chronic. Variations in genetic background, hypertension etiology, pharmacodynamics and pharmacokinetics may explain the different blood pressure responses to exercise among hypertensive patients. The present review discusses the different pathophysiological aspects of the response to exercise in hypertensives, including its modulators and diagnostic and prognostic usefulness, as well as the latest guidelines on prescribing and monitoring exercise regimes and drug therapy in the clinical follow-up of active hypertensive patients.
A actual sociedade urbanizada apresenta níveis manifestamente insuficientes de actividade para manter os níveis de saúde desejáveis, e prevenir, entre outras, as doenças cardiovasculares. O exercício aeróbico é um adjunto favorável à medicação no tratamento da hipertensão arterial, sendo desprovido virtualmente de efeitos secundários. Existem numerosos mecanismos propostos para o efeito salutar do exercício na redução da pressão arterial, sendo que os efeitos fisiológicos decorrentes podem ser classificados em agudos, pós-esforço, e crónicos. Diferenças no património genético, na etiologia da hipertensão, na farmacodinâmica e/ou farmacocinética podem justificar a diferente resposta tensional ao esforço entre os indivíduos hipertensos. Na presente revisão iremos abordar os vários aspectos fisiopatológicos relacionados com a resposta tensional ao esforço e seus moduladores no indivíduo hipertenso, sua utilidade diagnóstica e prognóstica, assim como referir-nos às últimas linhas de orientação no que respeita à prescrição/monitorização de regimes de exercício físico e fármaco-associação no seguimento clínico do hipertenso activo.
The increasing urbanization of modern society has led to profound changes in behavior, in particular a growing trend towards sedentary lifestyles. It is estimated that children nowadays expend 600kcal/day less in physical activity than their counterparts 50 years ago,1 while 57% of the population of Europe do not take regular vigorous exercise2 and only 11% of Portuguese believe that exercise significantly influences health.3
However, there is clear evidence of an inverse dose–response relation between total quantity and intensity of exercise and cardiovascular mortality, in both healthy individuals and in those with cardiovascular disease (CVD).4,5
It is important to differentiate physical activity from exercise and sports6 (Table 1), but all three are useful at all stages of the natural history of CVD.
Key concepts.
| Physical activity – body movements resulting in contraction of skeletal muscle, increasing energy expenditure above baseline levels |
| Exercise – planned, structured and repetitive body movements, for a specific purpose |
| Sport – activity requiring a range of physical abilities and vigorous physical exercise |
Exercise therapy is recommended in all the main clinical guidelines on CVD,7–9 and if properly implemented, it also has beneficial effects on risk factors for CVD, including hypertension, dyslipidemia, insulin resistance, obesity, and inflammation.10,11
In the case of hypertension in particular, the contribution of non-pharmacological treatment is central to reducing cardiovascular morbidity and mortality. This article reviews the mechanisms of the response to exercise and its modulators, the main benefits and risks involved, guidelines for prescribing exercise in hypertensives, and specific aspects of drug therapy in active hypertensive patients.
Acute physiological response to exerciseThe acute physiological response to exercise, during which the organism is subject to significant stress, varies in intensity according to the type of exercise, participant and environment. Exercise always causes transient organ dysfunction and generation of free radicals, but acquired adaptation mechanisms will improve functional performance in the future.
During endurance exercise there is typically an increase and redistribution of cardiac output (CO) to maintain perfusion of active muscles. This response is triggered by neurohormonal and hydrostatic mechanisms, initially by increasing systolic volume and then increasing heart rate.11 Systolic and diastolic blood pressure (BP), which are determined by the product of CO and peripheral vascular resistance (PVR), react differently to exercise: systolic BP (SBP) rises as CO increases, while diastolic BP (DBP) falls as a result of decreased PVR, facilitating perfusion of large muscle groups.
In resistance exercise both SPB and DBP rise as a result of the exercise pressor reflex, in which intramuscular pressure during contraction exceeds arterial pressure, interrupting muscle blood flow, and arterial pressure rises to overcome this resistance to muscle perfusion. If inappropriately applied, this type of exercise can cause SBP to rise up to 320mmHg and DBP up to 250mmHg during a single repetition at maximum load.12
Post-exercise physiological responseAn acute fall in BP minutes or hours after exercise,13 known as post-exercise hypotension,14 is seen in hypertensive and pre-hypertensive subjects15 and also in normotensive individuals, although to a lesser degree.16 It can last up to 22h after exercise.12 The principal mechanism suggested for this fall is reduced noradrenaline levels and thus inhibition of sympathetic activity and reductions in circulating angiotensin II, adenosine and endothelin and their receptors in the central nervous system, all of which reduce PVR and increase baroreflex sensitivity.17 The vasodilator effect of prostaglandins and nitric oxide released during exercise also contributes.17
This fall in BP is clinically significant, particularly in hypertensives, since it can act as a non-pharmacological BP-lowering measure. However, several aspects of the phenomenon remain unclear; various factors influence the hypotensive response, including the intensity, duration and type of exercise and the individual's clinical status, age, ethnicity, and physical fitness. Studies directly comparing different exercise intensities indicate that the fall in BP is independent of intensity in both normotensives and hypertensives.17 However, exercise duration appears to influence both the magnitude and the duration of the fall,17 although the volume of exercise appears to be more important than the action of other variables in isolation, with longer but less intense periods of exercise producing the same results as shorter and more intense sessions.17 This means individuals can exercise whatever their clinical status, including hypertensives, in whom the intensity should be carefully controlled. However, post-exercise BP response appears to differ with ethnicity: BP in black hypertensive women does not necessarily fall after exercise.17 Age may also modulate exercise response: PVR increases naturally with age, resulting in a smaller fall in BP. There are also indications that intermittent exercise protocols, and those that use more muscle mass, may increase the BP-lowering effect.
Hypertensive response to exerciseAlthough resting blood pressure measurement in the physician's office is the gold standard for management of hypertension, it is no longer considered the best tool to assess BP. Other methods provide more information on the risk associated with high BP, including 24-h ambulatory blood pressure monitoring (ABPM) and exercise testing, which correlate more closely with, for example, left ventricular hypertrophy (LVH) than office measurement.18
BP response to exercise is particularly useful in prognostic stratification of hypertensive patients as well as for diagnosing silent CVD. An abnormal response, in which SBP fails to rise or even falls during exercise, is universally accepted as a sign of severe ischemia, while a disproportionate hypertensive response must be taken into consideration when assessing the natural history of the disease.
A hypertensive response to exercise in sedentary subjects is defined as SBP rise of over 7–10mmHg for every increment of one metabolic equivalent (MET), or failure of DBP to fall more than 15mmHg (or actually to rise). Such a response to exercise is a strong predictor of risk of developing sustained hypertension19 and individuals with this response also have a higher rate of fatal cardiovascular events.20 A prospective study showed that cardiovascular mortality up to 16 years after exercise testing in 2000 apparently healthy individuals aged between 40 and 59 years was up to twice as high in those whose peak SBP was >200mmHg, and the risk was higher the steeper the BP rise during exercise. In fit individuals and those undergoing training, BP rises more slowly than in those in poor cardiorespiratory condition, in whom BP rise is rapid, steep and large immediately after beginning, or even before, exercise.21
Similarly, probably also due to adrenergic hypersensitivity, individuals with white coat hypertension usually have higher peak BP with exercise and are more likely to have LVH and lower nitric oxide bioavailability than the general population.22 Exercise BP correlates well with ABPM, to some extent reflecting circadian variations in BP. There is evidence that intermittent BP rises with exercise in these individuals may be sufficient to cause target organ dysfunction in susceptible and unfit individuals.23
The post-exercise response profile also has prognostic implications. A delayed post-exercise SBP fall, with a ratio of SBP at 3min of recovery to SBP at 1min of recovery of >1, is associated with worse prognosis.23
Chronic response to exercise: benefits and risks of physical activityAerobic exercise has been shown to reduce the incidence of hypertension and to lower BP in hypertensives, with SBP falling by 5–15mmHg (less in the elderly).24–27 Falls in DBP with this type of exercise, although beneficial, are less marked. The overall reduction is directly proportional to the duration and frequency of exercise28; it appears to be independent of body mass index,29 and is greater in middle-aged hypertensives,27 uncontrolled hypertensives, and non-dippers.28
The largest meta-analysis to date on the effects of aerobic exercise on BP, by Whelton et al.,30 analyzing 53 randomized controlled trials lasting over 2 weeks in individuals aged over 18, estimated SBP reduction as 5mmHg at most. Compared to previous meta-analyses which showed more dramatic reductions, this value is probably closer to reality, which may be due to the inclusion of longer trials with an intention-to-treat design, in some of which exercise regimes were not supervised, thus reflecting more realistic levels of adherence.
Resistance exercise has also been shown to reduce both SBP and DBP by 3–5mmHg31 by increasing parasympathetic tone and thereby reducing PVR32 when the exercise regime concentrates on muscle resistance.
Given that the fall in BP following both endurance and resistance exercise is relatively small,33 it is clear that exercise does not cure hypertension. Even so, it is an excellent adjunctive therapy and can alter the natural history of the disease. In middle-aged individuals, a decrease of only 2mmHg in SBP reduces cardiovascular mortality by 4%.34 The fact that exercise has pleiotropic protective effects, including reducing body weight, visceral adiposity and inflammation, and improving endothelial function, explains its overall benefit on cardiovascular risk profile, independently of reductions in BP. Increased physical activity and cardiorespiratory capacity can have a similar effect to drug therapy, reducing LVH by 8–15%,35 and to other non-pharmacological measures that reduce BP by 1–4mmHg.36
The mechanisms put forward to explain the BP-lowering and cardioprotective effects of regular exercise are based on neurohormonal and structural adaptations in vessels, muscle and adipocytes. The neuroendocrinological factors involved include reductions in circulating noradrenaline and its receptors and in angiotensin II, and increases in nitric oxide bioavailability,37 antioxidant capacity, insulin sensitivity, and expression of cardioprotective factors such as apelin.38 Structural adaptations include vascular remodeling (increased length and lumen diameter and number of precapillary sphincters) and neoangiogenesis.
Despite all the recognized benefits of exercise as a complementary treatment for hypertension, it is not free of risk in certain cases, and thus appropriate medical screening is essential before participating in exercise or sports.
In general, the main concerns are:
- a)
to identify absolute contraindications to particular types of exercise or sports;
- b)
on the basis of cardiovascular risk stratification, to identify limitations in the practice of particular types of exercise or sports (relative contraindications);
- c)
to tailor the exercise regime to each clinical condition in order to minimize complications and to promote rehabilitation (exercise medicine).
For hypertensives, the specific concerns are:
- a)
to diagnose silent CVD;
- b)
to identify individuals at high cardiovascular risk, such as those with uncontrolled stage 2 hypertension, unstable angina or decompensated diabetes, who should not exercise until their clinical condition has been stabilized;
- c)
to prepare a personalized plan designed to avoid activities that will tend to raise BP.
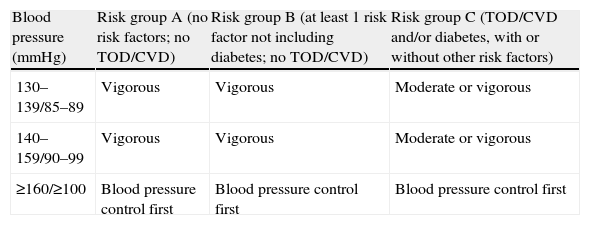
The most feared complication is sudden death, which in individuals aged over 35 is usually due to silent ischemic heart disease, although the former is uncommon in those followed medically. The thoroughness of pre-participation screening depends on the intensity of the planned exercise and the hypertensive patient's global cardiovascular risk. For an asymptomatic individual in risk group A or B39 with BP of <180/110mmHg intending to participate in light to moderate exercise (<60% VO2 reserve), there is generally no need for diagnostic exams beyond the routine evaluation14 (Table 2). Those in risk group C, without CVD or BP >180/110mmHg, may benefit from exercise testing before engaging in moderate-intensity exercise (40–60% VO2 reserve), but not for light activity (<40% VO2 reserve).14 Exercise testing is essential for all patients with documented CVD, whatever the level of intensity, and vigorous exercise (>60% VO2 reserve) should only be performed in dedicated cardiac rehabilitation centers.15
Algorithm for exercise testing before participation in exercise programs.
| Blood pressure (mmHg) | Risk group A (no risk factors; no TOD/CVD) | Risk group B (at least 1 risk factor not including diabetes; no TOD/CVD) | Risk group C (TOD/CVD and/or diabetes, with or without other risk factors) |
| 130–139/85–89 | Vigorous | Vigorous | Moderate or vigorous |
| 140–159/90–99 | Vigorous | Vigorous | Moderate or vigorous |
| ≥160/≥100 | Blood pressure control first | Blood pressure control first | Blood pressure control first |
Adapted from Ref. 14. CVD: cardiovascular disease; TOD: target organ damage.
The optimal exercise regime for hypertensives in terms of type, frequency, intensity and duration has been the subject of considerable research.
Type. Most studies focus on endurance exercise. As shown above, rhythmic aerobic exercise involving the main muscle groups, such as walking, running, cycling and swimming, reduces BP by 5–15mmHg. The response to resistance training is less marked (5mmHg), but it can be greater in circuit training, which involves more repetitions of lighter loads,40 since DBP rises more with greater static work, which should therefore be kept to a minimum. A resistance training program should set limits of >20mmHg over baseline DBP or DBP >120mmHg, and if these are exceeded the program and/or medication should be reviewed. The individual's preferences should also be taken into consideration, as this will affect long-term adherence.
Frequency. Exercise on 3–5 days a week reduces BP.41 Although there is evidence that 7 days a week may be more effective,42 three sessions a week have 75% of the antihypertensive effect of seven sessions.43 The simpler the regime the better, and it is not necessary to exercise every day to obtain an antihypertensive effect, especially as this effect lasts for many hours.
Intensity. Exercise intensity of less than 70% of VO2 reserve appears to have a more marked BP-lowering effect27 that does not depend on improved maximal aerobic capacity (VO2max). The main health benefit thus derives from changing a sedentary lifestyle to one of physical activity, with moderate intensity exercise being safest and most effective, as well as most likely to be adhered to. Reductions in BP obtained with intensities between 40 and 70% VO2max are similar.43 This intensity range corresponds to approximately 12–13 on the Borg 6–20 scale. Use of the Borg scale to monitor exercise intensity is of particular importance in patients taking beta-blockers, since these may weaken the hemodynamic response to exercise.
Screened individuals with low cardiovascular risk may benefit from interval exercise, with periods of varying intensity in a single session, since as well as leading to greater reductions in BP44 than with continuous exercise, this regime protects against age-related sarcopenia and loss of fitness.
Duration. Most randomized controlled trials to date in hypertensives have used continuous exercise lasting 30–60min per session. These times lead to similar BP reduction, but exercise programs of over 2.5h a week give very little extra benefit.32 The alternative of shorter intermittent sessions appears to be valid, since these elicit the same kind of BP response,45 so long as the volume of exercise is similar.
To summarize, exercise is recommended as a treatment for hypertension, particularly cardiovascular exercise,46 for 20–60min 3–5 days a week, at an intensity of 40–70% VO2max. Although resistance exercise in the form of muscle endurance training is no more effective in reducing BP, it may be integrated into the exercise regime so long as DBP response remains within safe limits.
Modulators of response to exerciseAlthough regular moderate exercise lowers BP in most hypertensives, not all respond to exercise therapy in the same way.34 Differences in genetics, pathophysiology of hypertension, pharmacodynamics and pharmacokinetics may explain why some individuals do not respond to exercise with falls in chronic BP values.
Rankinen et al.47 examined the association between Glu298Asp variants of the NOS3 gene, which codes for nitric oxide synthase, and BP response during submaximal exercise in 471 normotensive individuals after an endurance training program. Individuals with the NOS3-Asp allele reduced DBP less during submaximal exercise than those homozygous for the NOS3-Glu allele.
Hagberg et al. also reported differences in training-based BP reductions in hypertensive subjects with different alleles for the apoE gene.48 The endothelial expression of endothelin-1 promotes vasoconstriction, and certain variants of the endothelin-1 gene are associated with hypertension.49 Interestingly, the association between endothelin genotype and blood pressure phenotype is influenced by different levels of physical activity and functional capacity. In the HERITAGE Family study,50 20 weeks of endurance training at submaximal effort in sedentary individuals reduced SBP and pulse pressure less in carriers of the rs5370 allele than in homozygotes.
Regular exercise prevents or reduces age-related arterial stiffness. Besides its vasomotor effect, endothelin-1 is involved in vascular remodeling. Iemitsu et al.51 examined different genetic polymorphisms in the ET-A and ET-B receptors and in the two isoenzymes of endothelin-converting enzyme (ECE-1 and ECE-2) and their relationship with exercise-induced effects on age-related arterial stiffness. Individuals with the AA genotype of the 958A/G polymorphism of ET-A, and those with the AG or GG genotype of the 831A/G polymorphism of ET-B, presented a reduction in arterial stiffness with greater physical activity, while others showed no improvement with exercise, showing that exercise alone may not be sufficient to reduce age-related arterial stiffness in these individuals.
Monitoring of BP response to exerciseABPM has improved cardiovascular risk stratification and evaluation of individual exercise response, complementing conventional exercise testing. ABPM helps quantify the dose response to exercise, analyze circadian BP variations according to time of day for exercise, compare different exercise regimes, and divide exercise sessions into shorter periods.
As pointed out above, not all hypertensives respond in the same way to exercise; around 25% show no BP fall,27 although they still derive other benefits. Non-dipper hypertensives have been identified as being among those who do not respond to exercise.52 Non-dippers suffer more cardiovascular complications and more severe target organ damage, and would therefore seem to require more carefully planned and individualized exercise programs. However, on the basis of ABPM data, Park et al.53 concluded that non-dippers do in fact respond to exercise, but differently from dippers, in a way that depends on the time of day exercise is performed:
- 1)
Evening exercise appears to be more effective in reducing nighttime BP for non-dippers than for dippers.
- 2)
Morning exercise produces similar daytime SBP reductions for dippers and non-dippers.
- 3)
Morning and evening exercise exhibits similar 24-h SBP reduction for both dippers and non-dippers.
Time of day for exercise thus appears to be a useful concept in tailoring exercise regimes for non-dipper hypertensives.
There is also evidence that intensive training programs, in which patients’ response is more closely monitored, are better at motivating patients to modify their lifestyle, not only leading to greater falls in BP, but also improving their overall metabolic profile.54 Participation in programs in exercise centers for secondary prevention therefore seems an excellent option.
Drug therapy in active hypertensivesWhen drug therapy is indicated in active hypertensives, it should ideally: (a) lower BP at rest and during exertion; (b) decrease PVR; (c) not adversely affect exercise capacity. For these reasons, angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), and calcium channel blockers are the drugs of choice for hypertensive recreational exercisers and athletes. Due to the vasodilator effect of calcium channel blockers, they may cause post-exercise hypotension, and therefore an extended cooling-down period is recommended rather than abrupt cessation of activity. If a third drug is required a low-dose thiazide diuretic may be added, although the possibility of iatrogenic hypocalcemia or hypercalcemia should be monitored, as these could cause malignant arrhythmias during exercise.
Diuretics and beta-blockers can affect thermoregulation and increase the risk of hypoglycemia. Loop diuretics cause volume depletion, with severe consequences for exercise capacity. Furthermore, use of beta-blockers, besides their negative chronotropic and inotropic effects (which affect maximum exercise capacity), may also be considered as doping in some competitive sports. Non-selective beta-blockers can also impair normal bronchomotor tone during exertion. There is disagreement concerning their effects on intermittent claudication in patients with peripheral arterial disease55 and hence on maximum walking distance, and so they should be prescribed with caution when clinically indicated. Their use in active hypertensives should be restricted to cases of adrenergic hypertension or those with concomitant ischemic coronary artery disease; in the latter case ivabradine, when recommended in the guidelines, may enable better performance.56
There is no evidence of negative effects of any drug classes on static exercise.
ConclusionsThe adoption of a healthy lifestyle, of which exercise is a key element, is recommended for the treatment and prevention of hypertension. Programs that include endurance and resistance training not only play a part in the primary prevention of hypertension but also lower BP in hypertensive individuals.
The immediate effect of aerobic exercise is to increase and redistribute cardiac output, raising SBP while DBP remains the same or falls slightly, while both SBP and DBP rise during predominantly static exercise. Post-exercise effects in the 72h following an exercise session include a slight reduction in BP, especially in hypertensives (post-exercise hypotension), the fall being greater for higher baseline levels. Chronic effects result from the organism's adaptation to frequent exercise. Individuals who take regular exercise have lower blood pressure, relative bradycardia at rest, muscular hypertrophy, physiological left ventricular hypertrophy and increased oxygen consumption.
Decreased serum catecholamine levels and adiposity, increased insulin sensitivity and alterations in the expression of vasoconstrictor and vasodilator factors are among the proposed mediators of BP response to exercise. Differences in genetics, pathophysiology of hypertension, pharmacodynamics and pharmacokinetics may explain why some individuals do not respond to exercise with falls in chronic BP values.
In view of the added safety provided by pre-participation sports screening, there is justification for widening prescription of exercise programs to all hypertensives based on the results of such screening, since the benefits of exercise undeniably outweigh any complications that might occur.
The training program should be carefully designed and tailored in order to optimize its antihypertensive effects and to maximize safety. The following standard recommendations for exercise regimes for hypertensive individuals are proposed:
- a)
Frequency: on most, preferably all, days of the week.
- b)
Intensity: moderate (40–70% VO2R), in order to maximize the benefit and minimize the possible adverse effects of more vigorous exercise.
- c)
Duration: at least 30min of continuous or intermittent activity (3×10min) per day.
- d)
Type: primarily endurance exercise supplemented by resistance exercise.
The individual's preferences should also be taken into consideration, as this will affect long-term adherence.
First-line drug therapy should be ACE inhibitors or ARBs, possibly associated with thiazide diuretics if required. Loop diuretics and beta-blockers have secondary effects that can affect sporting performance and their use should be on a case-by-case basis.
In conclusion, aerobic exercise is a useful adjunctive therapy in treating hypertension, reducing cardiovascular and metabolic risk, and is almost completely free of secondary effects. It should be recommended to all hypertensive individuals who are willing and able to participate.
Conflicts of interestThe authors have no conflicts of interest to declare.
Please cite this article as: Ruivo JA, Hipertensão arterial e exercício físico. Rev Port Cardiol; 2012. doi:10.1016/j.repc.2011.12.012.